Mercury and Parkinson's Disease:
Mercury is toxic to the human body and specifically to motor neurons which when inflamed, disrupted, disabled or killed off by mercury, is a possible causative factor for Parkinson's disease. Mercury and many other heavy metals (lead, chromium, ...) are bio-accumulative, meaning that they make their way into the body, but the body can not easily dispose of it, thus accumulating over time. The primary human target areas susceptible to mercury are the brain, nervous system, kidneys, and the cardiovascular system. Mercury sources, measurement and detoxification by chelation (pronounced "key lay shun") therapy are all discussed.
Mercury can enter the body in three forms:- Elemental mercury (Hg): gas or liquide. Liquid state at room temperature. Gaseous state is particularly dangerous as about 80% of inhaled vapors are absorbed by the lung tissues. Elemental mercury is poorly absorbed through skin and GI tract but it is highly fat soluble allowing it to pass through cell membranes and the blood-brain-barrier. Once embedded in the brain or organs (binds to structural proteins), it can disrupt normal function. Used in thermometers, thermostats, latex paint (prior to 1991) and "silver-mercury" amalgam dental fillings (50% elmental mercury).
- Organic mercury (MeHg, EtHg or CH₃HgX): produced when elemental mercury is methylated by intestinal and oral bacteria. Combines with carbon to form organic molecules: methyl-mercury (very toxic), ethyl-mercury (EtHg), dimethylmercury, phenyl mercury. Methyl-mercury (MeHg) is a very dangerous form of mercury as it is easily absorbed by the GI tract. MeHg can bind to the sulfur-Hydrogen (SH) group of cysteine to cross the blood-brain barrier to disrupt glial cells and neurons in the brain. Organic mercury is also fat soluble allowing it to pass through cell membranes and the blood-brain-barrier. It is primarily excreted in feces but has a very low excretion rate and thus can have significant life long accumulation. Urine tests are not a reliable indicator of the level of organic mercury in the body. Organic mercury is typically detectred in blood tests for mercury by measuring ethyl-mercury. Organic mercury is found in fish (methyl-mercury), industrial chemicals and thimerosol (banned from US childhood vaccines in 2001) (ethyl-mercury).
- Inorganic mercury (Hg(II), HgCl₂, HgO, ...): produced when elemental mercury oxidizes and forms bonds with elements (eg chlorine, sulfur or oxygen) other than carbon. Also known as "reactive gaseous mercury", "divalent mercury" or "alkyl-mercury". It's not fat soluble so it can not pass into cells or the brain but can form strong bonds with enzymes and tissue making it difficult to remove once attached. Inorganic mercury is found in cosmetic skin whiteners, fungicides (including wood preservatives), pesticides and antiseptics. It can be formed by metabolizing methyl-mercury. Inorganic mercury tends to concentrate in the kidneys and is not known to cross the blood-brain-barrier.
The brain is one of the main targets for both elemental and organic mercury where it can remain for a lifetime. The most common toxic form is Methyl-mercury which is readily absorbed into the body through the gastrointestinal tract and has entered the aquatic food chain, thanks to the output from coal burning plants, eventually making its way into the oceans. Mercury has been shown to be a neurotoxin and can cause a large influx of calcium ions into the neuron cell, thus inhibiting dopaminergic neurotransmission which causes the physiological characteristics of Parkinson's disease (tremors, insomnia, muscle atrophy, muscle twitching, memory loss and degraded cognitive function) and even neuron cell death.
Sources of mercury include:- dental amalgam silver-mercury fillings: inhaled mercury vapor
- fish, especially carnivorous fish at the top end of the food chain or those with long life spans: shark, sword fish, halibut, orange roughy, king mackerel, tuna, marlin, bluefish, grouper
- thimerosol: a vaccine preservative, an anti-fungal agent, antibacterial preservative for antiseptic ointments, creams, jellies, nasal sprays, eye drops, contact lens solutions
- tattoo ink
- broken CFC light bulbs, fluorescent lamps, neon signs, mercury vapor lamps
- broken mercury thermometers, barometers, blood pressure meters
- mining: used to remove gold from ore
- electrical switches and motion detectors
References:
- Mercury is present in neurons and oligodendrocytes in regions of the brain affected by Parkinson’s disease and co-localizes with Lewy bodies (Pamphlett and Bishop, 2022) study based on autopsies of brain tissue measured with Autometallography and Laser ablation-inductively coupled plasma-mass spectrometry (LA-ICP-MS)
"Key findings of this study are that people with PD who have been exposed to mercury (ie, with locus ceruleus mercury) had mercury within neurons and oligodendrocytes in regions of the brain known to be affected by PD, and had mercury associated with a-synuclein aggregates in Lewy bodies and neurites."
"In our PD patients, neurons containing mercury were prominent in the substantia nigra, motor cortex, and thalamus, all regions with a decreased neuronal output in the classical model." - Mercury and Parkinson's Disease: Promising Leads, but Research Is Needed (Torret and Simmons, 2023)
"The brains from the individuals with Parkinson's disease had the heaviest and most widespread concentration of mercury, especially in brain areas such as the motor cortex, cerebellum, stratum, thalamus, and substantia nigra; in the last three of these areas, the two known Parkinson's disease brains were the only ones with mercury."
"The researchers speculated that the distribution of mercury in these two brains was also consistent with symptoms such as tremor, rigidity, and bradykinesia."
"Other studies have reported that individuals with Parkinson's disease have received more amalgam fillings than controls and that individuals who receive amalgam fillings are more likely to develop Parkinson's disease."
"Hg (mercury) and Pb (lead), which are of particular relevance to environmental-induced Parkinsonism, are among the most effective accelerators of alpha-synuclein fibrillation." - Parkinson's Disease as a Manifestation of Mercury Toxicity (Joe Bianco, 2020) an overview
- Parkinsonism in chronic occupational metallic mercury intoxication (Miller et.al.,2003)
- Methylmercury Induces Neurite Degeneration in Primary Culture of Mouse Dopaminergic Mesencephalic Cells (Gotz et.al.,2002)
- HEAVY METAL TOXICITY: LESSONS FROM A CASE OF SIMULTANEOUS OCCUPATIONAL EXPOSURE TO MANGANESE AND MERCURY
- Industrial toxicants and Parkinson's disease #Mercury
- Frontotemporal dementia and parkinsonism in a patient with a chronic mercury intoxication (Ciordia et.al., 2009)
- Epidemiologic study on the association between body burden mercury level and idiopathic Parkinson's disease (1989)
- World Health Organization (WHO): Mercury Exposure Guidance and Risks
- Mercury Toxicity and Treatment: A Review of the Literature (2012)
Negative Facts About Mercury:
- Mercury will accumulate in the lysosomes of motor neurons resulting in their dysfunction and destruction.
Note that lysosomes are the component of a cell responsible for alpha-synuclein recycling via autophagy, and when made dysfunctional by mercury, that leads to alpha-synuclein aggregation, Lewy body growth and the manifestation of Parkinson's.
References:- Heavy metal-induced disruption of the autophagy-lysosomal pathway: implications for aging and neurodegenerative disorders (2025, Das et. al.)
"As individuals age, the consequences of neurodegeneration become more significant, raising the likelihood of developing disorders like Alzheimer's and Parkinson's disease. This review explores the intricate relationship between heavy metal exposure, dysfunction of the autophagy-lysosomal pathway, ..." - Autophagy in Neurodegenerative Diseases and Metal Neurotoxicity (Ziyan Zhang et. al., 2016)
"PD is a late-onset neurodegenerative disease characterized by progressive and extensive loss of dopaminergic neurons in the substantia nigra pars compacta (SNpc) and the presence of aggregated a-synuclein-containing intracellular inclusions known as Lewy bodies. Lysosomal depletion and accumulation of autophagosomes are observed in the postmortem brain samples from PD patients, suggestive of defective autophagic clearance." - Accumulation of inorganic mercury in lower motor neurons of mice (1992, B. Arvidson)
- Heavy metal-induced disruption of the autophagy-lysosomal pathway: implications for aging and neurodegenerative disorders (2025, Das et. al.)
- Mercury also can bind to thiol (single sulfur group) containing molecules, such as cysteine which allows it to transported across the blood brain barrier.
References: - Mercury can cause tremors.
References:- Measurement of hand tremor induced by industrial exposure to metallic mercury (PDF) (Fawer et al, 1983)
"These neurophysiological impairments might result from the tendency of metallic mercury to accumulate in the cerebellum and the basal ganglia."
"The most important predictor variable of hand tremor was always the duration of exposure ( .. to mercury)" - Tremor in workers with low exposure to metallic mercury (Verberk et al, 1986)
"The study indicates that exposure to metallic mercury below the current TLV (50 micrograms/m3) may increase the tremor of the finger."
- Measurement of hand tremor induced by industrial exposure to metallic mercury (PDF) (Fawer et al, 1983)
- Mercury inhibits mitochondrial function.
References: - Mercury has an affinity for the brain
References:- The Toxicology of Mercury and Its Chemical Compounds (Clarkson, Thomas, Magos, Laszlo, 2006) (a rather complete introduction to mercury toxicity)
"The concentration in brain is about 5 times than in scalp hair about 250 times the corresponding concentration in blood"
"Animal data including primates indicate that brain levels in the newborn may be as high as five times the corresponding levels in the mother"
"Experimental work on rats has shown that methylmercury gains entry into the endothelial cells of the blood–brain barrier as a complex with cysteine (Kerper et al., 1992; Simmons-Willis et al., 2002)"
"Methylmercury is transported out of liver cells into bile as a complex with reduced glutathione using glutathione carriers (Ballatori and Clarkson, 1985; Ballatori et al., 1995)"
"The main route is via the feces, accounting for as much as 90% of total excretion according to animal observation"
"The inorganic mercury in brain appears to be virtually immobile, as substantially elevated levels have been found in autopsy brains of individuals who died at least 10 years after their last exposure to methylmercury (Davis et al., 1994)"
"As the dosage level of methylmercury rises to a certain critical level, the amount deposited in the brain sharply increases out of proportion to the increase in dose"
- The Toxicology of Mercury and Its Chemical Compounds (Clarkson, Thomas, Magos, Laszlo, 2006) (a rather complete introduction to mercury toxicity)
- Mercury eviscerates the neuron's dopamine D2 receptors making the neuron dysfunctional,
- Once embedded in the brain, mercury doesn't want to leave. Some areas of the brain are stickier than others.
No apparent half life was measured in the thalamus.
Reference:- A comprehensive review of mercury provoked autism (M. Geier, 2008)
"After dosing monkeys with organic Hg, ... six months after Hg dosing had ended, the concentrations of Hg2+ remained the same (thalamus) or doubled (pituitary)."
- A comprehensive review of mercury provoked autism (M. Geier, 2008)
The link between mercury and ill health including Parkinson's traits is clear and established.

Hg is derived from the Latin name for mercury: hydrargyrum
Mercury Toxicity Symptoms:
Note the commonality with Parkinson's symptoms. It's a Venn diagram of concentric circles.- fatigue, insomnia, depression, anxiety, nervousness, irritability, panic attacks, mood swings, emotional instability, fits of anger, shyness, withdrawal, fearful
- extreme mental distress, compulsion and depression leading to suicidal tendencies
- poor memory and cognitive function, dementia
- peripheral numbness, pricking sensations or tingling, decreased sense of touch
- slurred speech, insomnia, disrupted circadian rhythm, daytime sleepiness
- hypersensitivity, allergies, rashes, itchiness, eczema, psoriasis and other skin issues caused by mercury exiting the body via the skin
- chronic yeast or fungal overgrowth, constipation, malabsorption, irritable bowels
- physical tremors, ticks and muscle twitching, restless legs
- muscle weakness, muscle atrophy, muscle cramps, joint pain
- reduced motor coordination, difficulty walking, vertigo, balance (sensory nerve damage), dizziness, reduced visual-spatial awareness
- reduced (blurred) vision, tunnel vision, light sensitivity, difficulty focusing, poor night vision, poor color vision, poor hearing function, tinnitus
- tinnitus, hissing, high pitched ringing in the ear
- cardiovascular inflammation, atherosclerosis, heart disease, heart palpitations, elevated cholesterol and homocysteine, kidney disease, underactive thyroid, underactive adrenals, periodontal disease
- Diseases: fibromyalgia, Parkinson's, chronic fatigue, arthritis, crohn's, colitis, multiple sclerosis, alzheimer's, ADHD, Autism
- cellular dysfunction: interference with DNA transcription and protein synthesis, destruction of endoplasmic reticulum and ribosomes, alters mitochondrial function (exhaustion and fatigue)
- Inhaled mercury vapor: asthma, bronchitis, breathlessness, persistent cough, loss of sense of smell, chronic sinusitis, autoimmune diseases (multiple sclerosis, scleroderma, Hashimoto's thyroiditis, lupus), swollen lymph nodes
- Impairs cognitive function: poor memory, poor concentration, problems multi-tasking, indecision, lack of initiative, lack of motivation, difficulty finding words
- Reproductive dysfunction: infertility, low libido, severe period pains, miscarriages, still births, birth defects, low sperm count
Mercury and Neuron Toxicity:
Mercury is highly neurophilic (strong affinity for nervous tissue) and highly lipophilic (binds tightly to fatty tissue). Mercury is initially distributed by the circulatory system and enters nerve endings, traveling the central nervous system using a mechanism known as retrograde axonal transport leaving a path of destruction.
How Mercury Causes Brain Neuron Damage - University of Calgary
Note that mercury can cause neuron demyalation. Autoimmune driven demyalation is based on a different pathology and is typically suspect in neurological diseases such as muscular sclerosis, but not Parkinson's.
Correlation between Mercury and Parkinson's Disease:
Autopsies have found a correlation between mercury and Parkinson's Disease.
- Mercury is present in neurons and oligodendrocytes in regions of the brain affected by Parkinson’s disease and co-localizes with Lewy bodies - (2022)
"Mercury often co-localized with Lewy bodies and neurites."
"A more restricted distribution of brain mercury was seen in people without PD, with no mercury present in the substantia nigra, striatum, or thalamus." - Metals and Parkinson's Disease: Mechanisms and Biochemical Processes - [PDF] (Bjorklund et al., 2018)
"Parkinson’s disease onset has been associated with exposure to elevated levels of mercury. In fact, mercury has not only been associated with the incidence of PD, but several similarities between the effects of mercury exposure/ingestion, and the symptoms/consequences of PD have been identified"
"Low concentrations of Hg (1 mM) were able to abolish completely D2 dopamine receptors" (inhibits the flow of dopamine)
"testosterone synergistically enhances the toxicity of mercury"
"high levels of trivalent iron were found in Lewy bodies and dopaminergic neurons of the substantia nigra of PD patients"
DOI: 10.2174/0929867325666171129124616
Mercury And Parkinsonism:
Parkinsonism is the encompassing umbrella term that includes those diagnosed with the neurological symptoms of Parkinson's disease such as tremors, slowness of movement and rigidity. A person is diagnosed with Parkinson's when it is determined that they have the neurological disorder affecting dopamine flow and thus typically responds to lepodova medication. When a patient displays the symptoms of Parkinson's (eg. tremors, slowness of movement, rigidity, etc) but does not respond to lepodova medication, it is said that they have atypical Parkinson's or Parkinsonism but not Parkinson's. This condition is often caused by the side effects of medication, mercury toxicity, restricted blood flow in the brain, a brain tumor or traumatic brain injury. Note that the symptoms of mercury toxicity and Parkinson's are often indistinguishable and that neurologists rarely seek to diagnose root causes. While mercury may initially cause Parkinsonism and not Parkinson's, it will eventually cause Parkinson's due to its disruption of autophagy leading to the aggregation of alpha-synuclein and the formation of Lewy bodies.
Amalgam Dental Fillings:
Amalgam dental fillings are composed of about 55% elemental mercury, 44% silver and 1% of trace metals like zinc, copper and tin, which together, bond and form a solid metal material used to repair dental cavities. The United States Food and Drug Administration (FDA) still views the use of mercury amalgam fillings as safe and the American Dental Association (ADA) still regard mercury amalgam fillings as safe but research has shown otherwise. The Environmental Protection Agency (EPA) declared in 1988 that scrap dental amalgam material was a hazardous waste and OSHA has mandates on how to handle amalgam fillings. The act of chewing and brushing release mercury from dental fillings. Acids found in foods and drinks (especially if they are hot) can cause the release of mercury from the amalgam. It's not surprising that in 2008, Norway, Sweden and Denmark banned the use of mercury in dental fillings.
Typically the silver-mercury dental materials are shipped to dentists with the quantities in pre-measured containers ("precapsulated") so the quantity is "fool-proof" but the material quality and the mixing of the materials to form the amalgam is not and if not mixed completely with all mercury reacted with the silver, the amalgam will have the propensity to release the unbound mercury. This phenomena is more likely to occur at the greatest levels in the weeks following a dental restoration. Note that some dentists (typically not USA) may employ reusable capsules and may error on the ratio of the mix (a process referred to as trituration). This practice is old, subjected to exposure and oxidation, generally more error prone and time consuming and should be avoided. The mixing machines ("amalgamators") should oscillate at an optimum frequency, amplitude and duration for a given capsule length and size and at the optimum temperature, in order to form the best dental amalgam. Over-mixing creates an amalgam which is hard to form and porous and prone to fracture while under-mixing may create an amalgam which is too soft with too much free mercury. Dentists who manually mix amalgam will be prone to have an even greater risk of error.
In order to mitigate any problems with the release of mercury from amalgam dental filings, the fillings can be removed and replaced with composite or ceramic material. The tooth can also be given a crown or a root canal performed to replace the tooth with a prosthetic. Amalgam replacement must be done wisely as it has the potential to generate mercury vapor and fine particles which can be ingested, resulting is an even higher dose of mercury poisoning. Amalgam removal should not be performed on those are or about to be pregnant. It is essential to have a mercury safe dentist who follows specific safety protocols when removing the fillings.
Mercury: The poison in your teeth: Amalgam stimulated with a tooth brush (Dr. Tom McGuire DDS)
Research has shown the relation of exposure to dental amalgams and the toxic burden of mercury in the human brain and kidneys.
References:- Mercury concentrations in the human brain and kidneys in relation to exposure from dental amalgam fillings (Nylander et al, 1987)
"It is concluded that the cause of the association between amalgam load and accumulation of mercury in tissues is the release of mercury vapour from amalgam fillings." - Study on the significance of mercury accumulation in the brain from dental amalgam fillings through direct mouth-nose-brain transport (Maas et al, 1996)
"a statistically significant correlation exists between the number of dental amalgam fillings and the Hg concentration in the kidney cortex, and, to a lesser extent, the Hg concentration in the occipital lobe cortex" - Human Exposure and Health Effects of Inorganic and Elemental Mercury (Jung-Duck Park, 2012)
"From autopsy studies, mercury levels in tissue samples including the brain were correlated with the total number of surfaces of amalgam restorations."
"... each 10 amalgam surfaces increased mercury level by 1 µg/L in urine in an association study on mercury levels in urine and blood and amalgam exposure." - The effects of amalgam restorations on plasma mercury levels and total antioxidant activity (Ozdabak et al, 2008)
"Plasma mercury (P-Hg) levels were found to be significantly higher in subjects with amalgam restorations when compared to subjects without amalgams"
DOI: 10.1016/j.archoralbio.2008.05.012 - Association between History of Dental Amalgam Fillings and Risk of Parkinson’s Disease: A Population-Based Retrospective Cohort Study in Taiwan (2016)
A Taiwan study found that people with amalgam fillings were 1.6 times more likely to get Parkinson’s than those without exposure to them regardless of sex or age. - Long-term Dissolution of Mercury From a Non-Mercury-Releasing Amalgam
Autopsies of people with dental amalgam fillings revealed that they have 2-12 times more mercury in their tissues than normal. It has been shown that the mercury in amalgam fillings releases mercury vapors which are inhaled and make their way into the body and brain.
Amalgams and Teeth Bleaching:
Little has been done to warn the public about this very serious hazard. Teeth bleaching with peroxide whiteners will trigger the release of mercury when in contact with silver-mercury amalgam dental fillings. The quantity of peroxide in the whitener, the surface area of the filling and the duration of treatment are important factors which affect the amount of mercury released. It has been shown that a silver colored, toxic slurry of mercury can be found in the "trays" (the mouthguard fixture used to hold the whitener in contact with the teeth) after a multi-hour whitening treatment. DON'T DO IT!
References:- Effects of In-Office and Home Bleaching Gels on the Surface Mercury Levels of Dental Amalgam (Oskoee et al, 2010)
"The tested bleaching products significantly elevated the surface mercury levels of amalgam in vitro."
Safe Mercury Amalgam Removal Technique:
The International Academy of Oral Medicine and Toxicology (IAOMT) has developed the Safe Mercury Amalgam Removal Technique (SMART) protocol recommendations. The IAOMT website also has a searchable worldwide dentist database or check out Dr. McGuire's database for a dentist with skills to meet the IAOMT protocol for removing amalgam fillings.
Safe protocols for amalgam removal include:- the use of a dam around the teeth being worked on to trap debris from being ingested by the patient
- the irrigation with cold water during removal to minimize the generation of mercury vapor
- the use of amalgam capture separators to capture material so that it is not released into the public waste system or atmosphere
- the use of an evacuation system to remove mercury dust and vapors from the patient's mouth
- the use of an air supply so the patient does not breathe in mercury vapors
- the physically coarse mechanical removal of chunks of amalgam rather than just the use of a fine drill which is more prone to generate fine particles of mercury, mercury dust and mercury vapor
- the use of ambient high volume air filtration equipment to trap mercury dust and vapors
- provide the patient a charcoal slurry binder (or equivalent) to rinse and swallow before and after the procedure
- provide the patient and dental workers protective gowns
- provide the dental workers protective face shields, head coverings, non-latex nitrile gloves and mask
- dentist should use equipment optimized to reduce aerosols, minimize and capture vapors and particles. For example the Zyris "Isolite" dentist drill minimizes aerosol and spatter, evacuates vapors and particles and isolates the work area.
IAOMT Safe Mercury Amalgam Removal Technique (SMART) protocol. Ignore the comment about taking chlorella (1 min and 3:53 min into the video) as it is not a good chelator (pronounced KEY-lay-tor) and will mobilize rather than remove mercury.
Amalgam fillings can be replaced with porcelain inlays or onlays bonded to the tooth or with a composite.
Be sure to use one of the newer bonding agents which do not contain Bisphenol A (BPA) which is a bio-accumulative, xenoestrogen endocrine disrupting chemical which has estrogen hormone like properties.
This is also true in choosing composite filling materials.
Choose materials which are BPA-free.
Also be sure to reduce your BPA exposure by choosing glass over canned food products and don't microwave polycarbonate plastic (marked with a number 7 recycling code) food containers.
Dental materials testing panel: Clifford Consulting and Research (get tested for BPA and other dental materials)
The goal of removing mercury amalgam dental fillings is to remove a source of mercury which will constantly release low doses into your body. Another reason is that it is a prerequisite for a mercury detoxification process known as chelation therapy which is used to remove mercury from one's brain and body tissue. It is a requirement to remove amalgam fillings prior to chelation as chelation will pull mercury from amalgam fillings creating a toxic event.
According to the ACC mercury detox protocol, any remaining bit of amalgam remaining in the tooth near the tooth root might induce the need for a root canal as chelation agents will be drawn up through the root of the tooth in an attempt to bind with the mercury. A grey stain remaining on the inside of a filling is more likely to be silver oxide and not mercury. A visible white spot revealed on an x-ray may be problematic and an indication that a speck of mercury amalgam is remaining in the tooth.
Binders such as activated charcoal can be taken 20 to 30 minutes before the amalgam removal procedure starts for the purpose of attaching to any mercury particles that may be ingested. For more on binders, see below.

Removing dental mercury amalgam using the SMART protocol (dental dam, respiration and evacuation equipment, protective garments, etc) Dr McBride DDS
For more details and photos see the IAOMT SMART protocol for mercury amalgam removal.
Dental organizations dedicated to mercury free dentistry and safe amalgam removal:- IAOMT: International Academy of Oral Medicine and Toxicology
- IABDM: International Academy of Biological Dentistry and Medicine
- HDA: Holistic Dental Association
Research has shown a significant reduction in the level of mercury found in blood plasma after amalgam removal.
- Mercury in biological fluids after amalgam removal (Sandborgh-Englund et al, 1998)
"An exponential decline of Hg was seen in all media. Sixty days after the amalgam removal, the Hg levels in blood, plasma, and urine had declined to approximately 60% of the pre-removal levels."
"After removal, there was a considerable decline in the Hg levels of blood, plasma, and urine, which slowly approached those of subjects without any history of amalgam fillings." - Mercury in saliva and feces after removal of amalgam fillings (L Björkman et al, 1997)
"In saliva, there was an exponential decline in the Hg concentration during the first 2 weeks after amalgam removal"
" Hg levels in all media decrease considerably after amalgam removal."
Chelation Therapy for the Removal of Mercury from Human Tissue and Organs:
Mercury accumulates in body tissue and organs and generally can not be removed mechanically. Be very skeptical of those offering a vitamin cleanse to remove mercury as there is no evidence that it will be effective. The act of using a mercury binding molecule to attach itself and engulf mercury atoms in a way that it is viewed by the body as organic waste that can be expelled by the body's regular organs, the liver (including bile and GI tract) and kidneys, is known as a chelation therapy. The goal of the chelator is to bind to the toxic metal to make it non-toxic, water soluble and easily excretable by the body. Mercury is known to to have a very high affinity to sulfhydryl (SH) groups. This affinity is exploited to attach to mercury and remove it.
The chelating agents possess a ligand (molecule that binds to a central metal atom by donation of one or more of its' electron pairs) that forms a bond with the metal to be removed. The chelating agent forms a stable complex with the toxic metal to shield human tissue from the metal ion, thereby reducing the local toxicity. The resulting molecule can then be mobilized and excreted in urine or feces.
Warning: While the chemistry of chelation is well known, there are not a lot of studies showing efficacy and many can do more harm than good if they mobilize rather than remove mercury or inflame the brain due to relocation of mercury to the brain. In most cases chelators will bond to mercury in organs and body tissue only to relocate it to the brain and central nervous system which have a high affinity and attraction to mercury, causing even greater harm to the patient. During one's lifetime, their body can accumulate a lot of mercury, enough to cause serious dysfunction if relocated from the body to the brain and nervous system which are much more sensitive to mercury. Attempts to continue chelation to clear the brain and nervous system of mercury often have the opposite effect due to further relocation. This website does not endorse chelation or any particular chelation therapy due to the dangers and extreme risks involved. Chelation is neither a reliable nor a safe therapy. Consider a safer option such as sauna therapy and sweating.
Chelation Agents:
| Chelation Agent | FDA | Description | Toxicity LD50 mmol/kg (lower = more toxic) | LD50 g/kg |
|---|---|---|---|---|
| DMSA | yes | Dimercapto Succinic Acid (DMSA) C4H6O4S2 is considered safer and less toxic than DMPS.
Goes by the trade name "Chemet" but also called "Succimer". DMSA does not enter the cells but relies on glutathione or other mercury mobilizer to push toxins out where DMSA is waiting to bind to them.
DMSA is typically given for 3 days followed by 11 "off" days. The body needs the "off" days to remake glutathione.
The dosage is based on body weight.
Chelates lead and mercury (favors organic ethyl-mercury).
DMSA targets extracellular (outside the cell) mercury, the mercury found outside the cell walls in body tissue.
DMSA does not have the ability to pass through cell membranes and must team up with glutathione or ALA to grab intracellular (inside the cell) mercury.
Not to be used when dental amalgam fillings are still resident as saliva delivery of the chelating agent will bind to the mercury and can result in acute mercury poisoning.
Digestible and does not result in a significant loss of essential metals like zinc, iron, calcium and magnesium.
Concurrent use with EDTA is not recommended.
Side effects include seizures, increased self-stimming, compromised central nervous system function, ataxia, convulsions, nausea, diarrhea, anorexia, headache, dizziness, sensorimotor neuropathy, decreased urination, arrhythmia, and infection.
FDA max adult dosage: 30 mg/kg/day 5 days, 20 for additional 14 days, 2 week break (ref).
DMSA was approved by the FDA in 1991.
Also see:
| 13.58 | 2.48 |
| MiADMSA | no | Monoisoamyl DMSA, C9H16O4S2, was synthesized by generating an ester of DMSA which is more water soluble and lipophilic (better able to disolve in fats) when compared to DMSA making it more capable of mobilizing intracellularly (inside the cell walls) bound toxic metals.
MiADMSA removes heavy metals from both intra and extra cellular sites.
Found to be more effective at chelation of mercury, arsenic, cadmium and lead without redistribution when combined with DMSA.
Use of DMSA with MiADMSA has been found more effective than mono-therapy with MiADMSA providing better clinical recoveries and minimize the possible side effects.
Co-admission with antioxidants such as vitamin C, E and Zinc reduces oxidative stress.
Also see:
| 3.0 | |
| MchDMSA | no | monocyclohexyl DMSA has a cyclic carbon chain and straight and branched chain methyl group similar to DMSA. Lipophilic compound with the ability to penetrate into cells. Can be given oraly. Can significantly reduce cadmium and arsenic levels. | ||
| MmDMSA | no | monomethyl DMSA has a cyclic carbon chain and straight and branched chain methyl group similar to DMSA. Lipophilic compound with the ability to penetrate into cells. Can be given oraly. Can significantly reduce cadmium and arsenic levels. | ||
| DMPS | no | 2,3-Dimercapto-1-Propane Sulfonic Acid (DMPS) C3H7O3S3Na.
Goes by the trade name "Dimaval".
Fast acting. May be over effective on first use.
Not to be used when dental amalgam fillings are still resident as saliva delivery of the chelating agent will bind to the mercury and can result in acute mercury poisoning.
DMPS favors inorganic mercury.
DMPS targets "extracellular" mercury, the mercury found outside the cell walls in body tissue.
DMPS can create toxic complexes with copper.
Avoid DMPS "challenge tests" as it creates a toxic mercury load.
DMPS is legal in Germany (1976) but not FDA approved for the USA. Also a strong chelator of copper.
Also see:
| 5.22 | 1.1 |
| EDTA | yes | Ethylene Diamine Tetra-Acetic acid (EDTA) amino acid was used in the National Institute of Health (NIH) TACT trial.
Considered safe by the FDA although it can be toxic to the kidneys.
Kidney function should be monitored. EDTA has the added benefit of clearing plaques in blood vessels.
EDTA does not form as strong a bond with mercry as do the di-thiol chelators.
EDTA was first used in the 1950's as a treatment for heavy metal (lead, mercury, copper, iron, calcium, chromium, cobalt, cadmium, magnesium, vanadium, zinc, ...) poisoning.
Best used for detoxification treatment of cadmium and lead and not mercury. Avoid "challenge tests" as it can create a toxic load.
According to Dr. Andy Cutler PhD, it can form a dangerous compound when it binds with mercury (hgEDTA) which is even more difficult to remove.
There are two versions, NaCaEDTA (calcium EDTA C10H12CaN2Na2O8) and NaEDTA (disodium EDTA) of which NaEDTA easily binds with calcium and can cause hypocalcemia induced cardiac arrest and thus should not be used on children.
Calcium EDTA, which is approved to treat lead toxicity can be given as a short IV push, but disodium EDTA, which may be used as a potential treatment for vascular disease, must always be given by a slow intravenous drip, at a rate no more than 1 gram per hour, otherwise, the calcium blood level drops dangerously fast.
| 10 | 4-6 |
| ALA | no | Alpha Lipoic Acid (ALA) C8H14O2S2 crosses the blood-brain-barrier and is used after chelation with other agents or in combination with other chelation agents.
ALA targets "intracellular" mercury, the mercury found inside the cell walls on both sides of the blood-brain-barrier (BBB).
Must wait three months after amalgam removal before starting chelation with ALA.
Many believe that ALA on its own is not an effective chelator but a compound which can support other chelators.
Excretion pathway: liver.
Half life: 3 hrs
Also see:
| ||
| Glutathione | Glutathione (GSH) is available as a neuro-protective supplement which is hard for the body to absorb and thus is more effective if given intravenously but is still ineffective due to a short half life of less than 20 minutes (ref).
It has been found to be far more effective to let the body produce GSH by consumption of its precursors: N-AcetylCysteine (NAC), Glycine and Glutamate.
Liposomal glutatione is an encapsulated form of glutathione which can easily be absorbed orally and pass through tissue and is the primary mercury mobilizer in Dr. Shade's mercury detox protocol..
Glutathione is also supported by a diet of sulfur-rich foods: garlic, ginger, onions, eggs, and broccoli sprouts.
Glutathione is typically generated by cells in the body as a natural detoxifying agent.
Mercury has an adverse effect on Red Blood Cell (RBC) glutathione levels, making it more difficult for a mercury toxic body to excrete mercury.
Glutathione is often not considered a true chelation agent because it does not contain two or more binding groups (dithiol groups), it has one.
ACC protocol considers Glutathione IV a dangerous option as it will redistribute mercury causing more damage rather than remove the mercury from the body.
Supplementing the precursors to glutathione, allowing the body to generate glutathione is preferred to intravenous solutions for those administering their own detoxification.
Glutathione has the ability to work intracellularly (inside the cell membrane) and can pass mercury to extracellular (outside the cell) chelators such as DMSA and DMPS.
Typically two glutathione molecules are required to bind and chelate a single mercury atom.
Glutathione excretion pathway: liver.
| |||
| Dimercaprol | yes | Dimercaprol C3H8OS2 (also called British Anti-Lewisite or BAL) is used to tread lead, mercury, bismuth, silver, nickel and arsenic poisoning. High doses can cause coma and seizures. Found to increase mercury levels in the brain. Not to be used to chelate iron or cadmium because the resulting complex is toxic. | 1.1 | 0.090 |
| DPA | yes | Penicillamine (DPA) C5H11NO2S sometimes used to treat bismuth, copper, lead, mercury, and nickel toxicity.
Primary adverse effect is an allergic reaction in people who are also allergic to penicillin.
Tends to redistribute mercury with adverse reactions.
D-penicillamine is only used for elemental and inorganic mercury toxicity and is not useful for organic mercury toxicity.
DMSA has replaced penicillamine, because of its strong metal-mobilizing capacity and lower side-effects.
Also see: The enigma of parkinsonism in chronic borderline mercury intoxication, resolved by challenge with penicillamine The d-penicillamine (dimethyl cysteine or (2S)-2-amino-3-methyl-3-sulfanylbutanoic acid) variant, also known as Cuprimine, is an oral chelator which is less toxic and preferred over the l-penicillamine variant. Also see:
| 0.337 | |
| DFO | yes | Deferoxamine (DFO) C25H48N6O8 mesylate is a chelator used to treat iron and aluminum (AL) toxicity. Primarily used for AL-related diseases in renal patients. | 0.520 | |
| SH-SAMMS | SH-SAMMS chelator which acts like a binder. Used to bind with mercury, cadmium and lead.
Not shown to detox the brain.
| |||
| Emeramide (NBMI, Irminix, OSR, BDTH2) | trial | N1,N3-bis-(2-mercaptoethyl) isophthalamide (NBMI) (chemical name) also known as Irminix (drug name), Emeramide (INN generic drug name) or OSR (oxidative stress relief when marketed as a dietary supplement) is a chelator and antioxidant which is hydrophobic and can theoretically pass through the blood-brain-barrier and chelate mercury from cell organelles like mitochondria. Emeramide/NBMI was commercialized by the biotech firm EmeraMed, passed FDA phase 1 and 2a human trials and was available for "compassionate use". NBMI is a di-thiol chelator which forms a strong bond with mercury (will not drop and mobilize mercury - known as the kinetic property of the chelator) and is excreted through the bowel in the stool. Once NBMI binds to mercury, the mercury becomes non-toxic. NBMI is excreted through the cytochrome P-450 (cytochrome: enzyme family, P: protein, 450: max wavelength of absorption in nano meters when enzyme is bound to CO) system pathway (a heterogeneous group of about 50 enzymes that catalyzes various oxidative reactions in the liver, intestine, kidney, lung, and central nervous system to support detoxification). The P450 enzymes break down xenobiotic compounds including NBMI but in no way does it manage the excretion of mercury. This is where NBMI fails. It fails to excrete mercury. Also note that some foods and supplements can inhibit the CY P-450 pathway (Mechanism of CYP2C9 Inhibition by Flavones and Flavonols). NBMI excretion is a slow process taking about 30 to 60 days to complete (ref). A study with rats showed no significant drop in tissue mercury burden within 5 days. Excretion of mercury in urine or feces was lower with NBMI within 5 days. NBMI can also bind to a lesser extent with Arsenic, Lead and Cadmium and not with essential minerals like Calcium, Magnesium, Potasium or Sodium. The manufacturer claims that Emeramide/NBMI is non-toxic and does not deplete essential minerals although Dr Haley does support zinc supplementation 3 hours before or after taking NBMI. It also discriminates bound metals so that it does not deplete essential iron and copper bound to proteins but will chelate free iron (Fe2+) and free copper (Cu2+). Note that free iron along with hydroxyl (OH) and other free radical species, has been implicated in the pathogenesis of Parkinson's disease. The order of NBMI affinity from strongest to weakest: mercury, lead, cadmium, gadolinium, thalium, coopper, zinc NBMI raises glutathione (GSH) levels by reducing the body's consumption of glutathione used to address toxic heavy metals (ref). NBMI is also a powerful antioxidant with an ORAC value of 192,400 (by comparison Acai fruit has an ORAC value of 18,500). Note: people with suspected yeast or Candida problems, or anyone who is on antifungal medications, should not take Emeramide/NBMI until their yeast conditions were under control. EmeraMed is developing Emeramide with PCI Pharma Services to provide a quality pharmaceutical grade and purity, supply. NBMI is also available as an industrial chemical which is NOT suitable for human consumption. Shelf life: 5+ yrs. Excretion pathway: liver. Half life: 18 - 22 hrs Videos:
| > 5 | |
| Cysteamine | yes | (HS(CH2)2NH2) Under investigation as a mercury chelator. Has a thiol group which may bond to mercury. FDA approved for the treatment of cystinosis, which is unrelated to its use as a chelator. Related to NBMI in that NBMI consists of two cysteamine molecules coupled to a single molecule of dicarboxybenzoate. Linear coordination of Hg(II) by cysteamine (Atwood et al., 2002) |
Chelation therapy must be avoided prior, during and immediately after dental mercury amalgam replacement (note: the developers of NBMI claim to be free of this problem). Do not consider chelation prior to dental amalgam removal as the chelation agent may be delivered by one's saliva to the mouth and dissolve the mercury in the amalgam resulting in acute mercury poisoning. Also note that dental amalgam replacement may temporarily elevate mercury in the body. Thus it is considered unwise to undergo chelation for four days to a week after amalgam replacement when using DMSA or DMPS or for three months after amalgam replacement when using ALA. One should find an IAOMT SMART certified dentist who has the safety gear to keep both the patient and dentist safe from mercury vapors and/or particles generated during the amalgam removal process.
All effective chelation protocols will call for the replacement of beneficial vitamins and minerals, which are also removed by the chelation agents.
The removal of mercury amalgam dental fillings with modern methods is a no-brainer as they can be safely removed without exposing the patient to harm. Chelation on the other hand does not have a clear and defined exit path for the mercury being removed and thus can impose harm on a patient, depending on the chelator. The mercury being removed from the body by chelation is often buried deep within tissues spread out among organs, fat and brain matter and the journey it takes to exit the body often includes being relocated after being dropped by the chelator compound and traveling through healthy tissue causing new mercury toxicity damage. Chelation requires balancing the type and dosage of the chelator so that the removal rate is tolerable, the toxicity of "droped" mercury is not inflammatory or hazardous, the burden on the kidney or liver excretion path is not doing organ damage and the accidental chelation of essential nutritional minerals is not detrimental. Chelation can present many hazards and requires recovery breaks for the body to heal. If chelation is causing pain, severe anxieties or depression, brain fog, head aches or discomfort, stop chelating as the dosage is too high! Chelation does not benefit the patient who is tough or is content with suffering as it may be doing damage. If the patient body burden of mercury is high, start light and be safe. The discussion of chelation therapies below is not an endorsement of chelation, any specific chelation agent or of any one chelation protocol. When one begins chelation therapy, they are starting at the highest level of mercury burden in the body and thus should start with the lowest dosage of chelator and stay at a low dosage for many rounds. The dangers of chelation are discussed in detail below.
Also see: Chelation in Metal Intoxication (2010) covers chelation agents, combinations of agents, pros, cons, limitations, etc
Chemistry of Dithiol Chelators:
The dithiol (two thiol organosulfur compounds of S-H, Sulfur-Hydrogen) group of chelators (DMSA, DMPS, ALA, NBMI, etc) work as chelation agents because they have two sulfur atoms available to bind with mercury (and drop a bond eg to Hydrogen), to form two bonds with the mercury atom. Note that the sulfur-mercury bond is strongest in a 180 degree bond and thus geometries which includes multiple molecules in a 2:2 ratio of chelator to mercury atoms is found to be more prevalent. Once the bonds with the mercury atom have been made, the mercury should no longer cause harm to the body and will expose an organic shell which the body can expel. In reality, mercury is often dropped and relocated causing new harm in new tissue.
DMSA: is a dithiol chelator which bonds to mercury and exits the body mostly via the kidneys and urine with small amounts (<1%) excreted by the liver, GI tract and feces. DMSA can not cross the blood-brain barrier but can rid the body of mercury and lower blood level concentrations of mercury. The resulting DMSA-mercury compound is excreted by the kidneys in urine, thus DMSA chelation must be accompanied with drinking a lot of water in order to purge the DMSA-mercury chelate. The result of not consuming enough water will be to retain mercury and to allow it the opportunity to be redistributed.
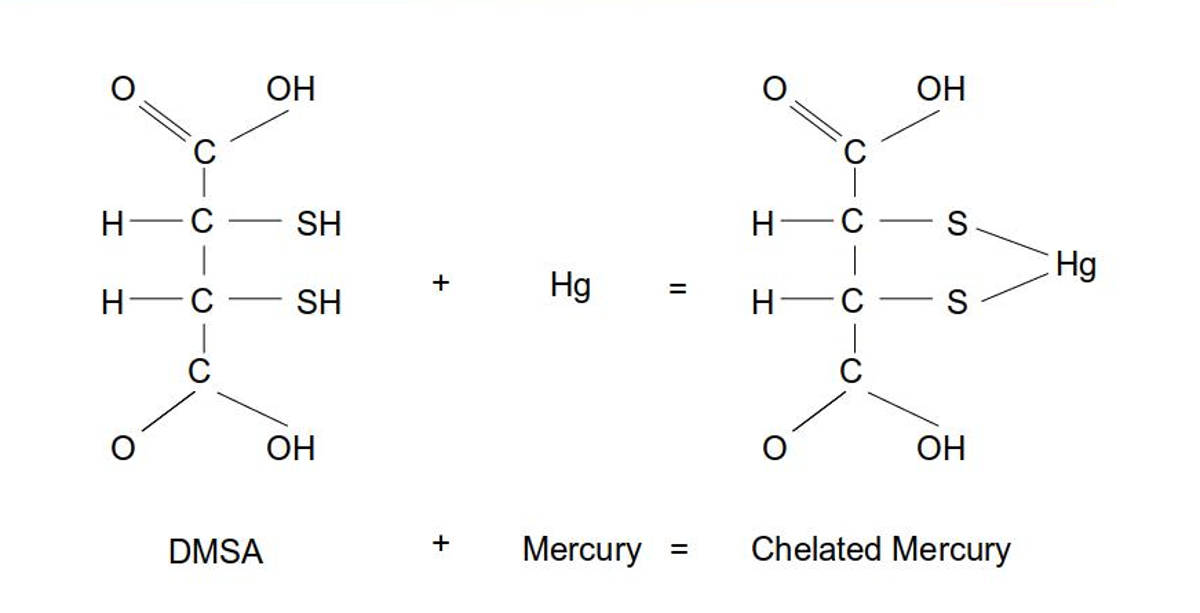
It was once thought that one molecule of DMSA bonded with one atom of mercury. Research by Graham George proved that DMSA and mercury do not bond in a 1:1 ratio.
Ratio DMSA:Hg shown is 1:1. Other ratios are more common (eg 2:2) with geometries supporting two 180 degree bonds with two mercury atoms.
A one-to-one ratio of DMSA bonding to one mercury atom tends to release the bond as the two sulfur atoms in the DMSA molecule are located on adjacent carbon atoms, not leaving sufficient space to reliably hold a mercury atom.
Thus DMSA prefers to bond to mercury using two DMSA molecules each with a single bond to a mercury atom resulting to a weaker bond than chelators such as the NBMI molecule which has the ability to form two stable bonds to a mercury atom.
This leads to the DMSA (and similar chelators such as DMPS and ALA) trait of dropping and relocating mercury.
For a study of DMSA configurations see Mercury binding to the chelation therapy agents DMSA and DMPS and the rational design of custom chelators for mercury (PDF) (George et al, 2004)
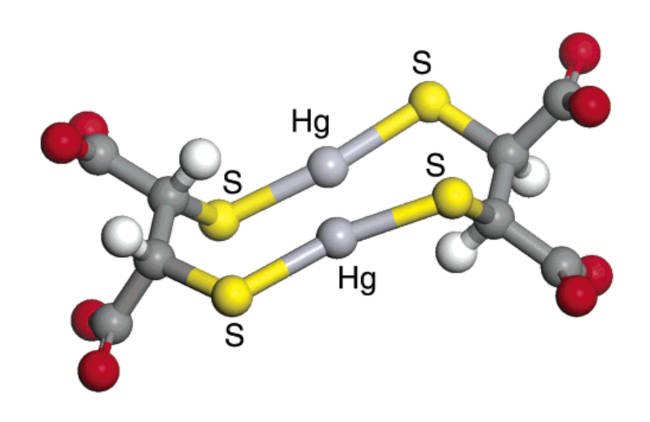
Research by Graham George shows that DMSA bonds with mercury in a 2:2 ratio
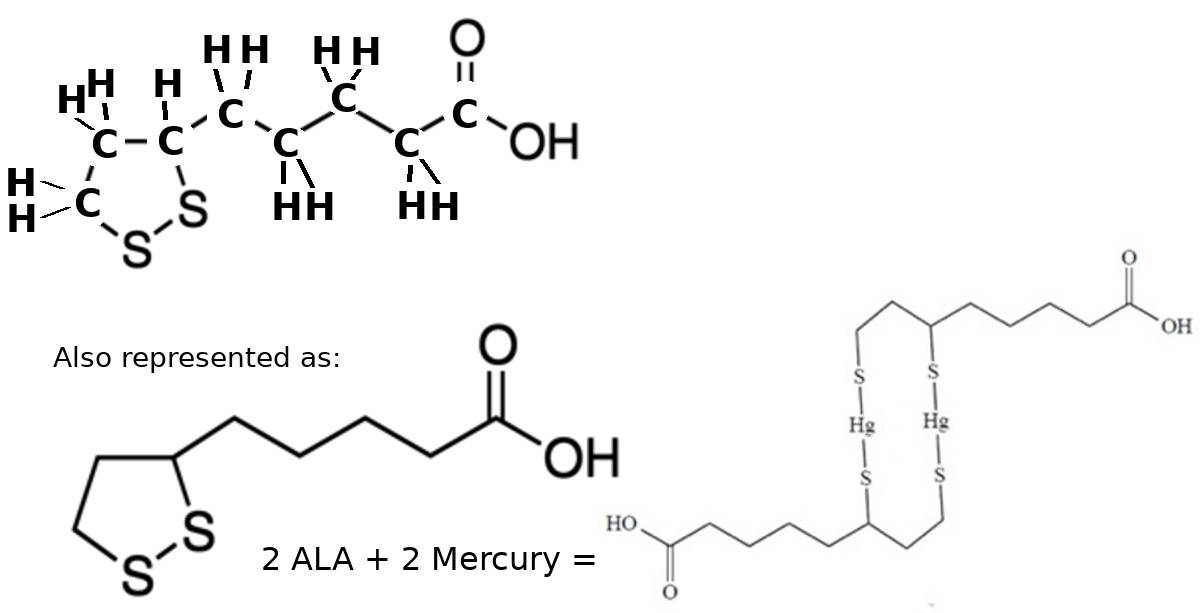
ALA: Similarly Alpha Lipoic Acid (ALA) has two sulfur atoms which can bind to mercury.
ALA can cross the blood-brain barrier to draw mercury from the brain (ref).
Preparation is required so that the natural flow will be from brain (area of high concentration) to the body (where the body concentration of mercury was lowered prior by chelation with DMSA).
According to Dr. Andrew Cutler's chelation (ACC) protocol, it is essential that this concentration gradient be established prior to using ALA as ALA is capable of moving mercury in and out of the brain.
Chelation of mercury (Hg) using Alpha Lipoic Acid (ALA) C8H14O2S2
ALA chelated mercury is typically excreted by liver in bile.
Ratio ALA:Hg is 2:2
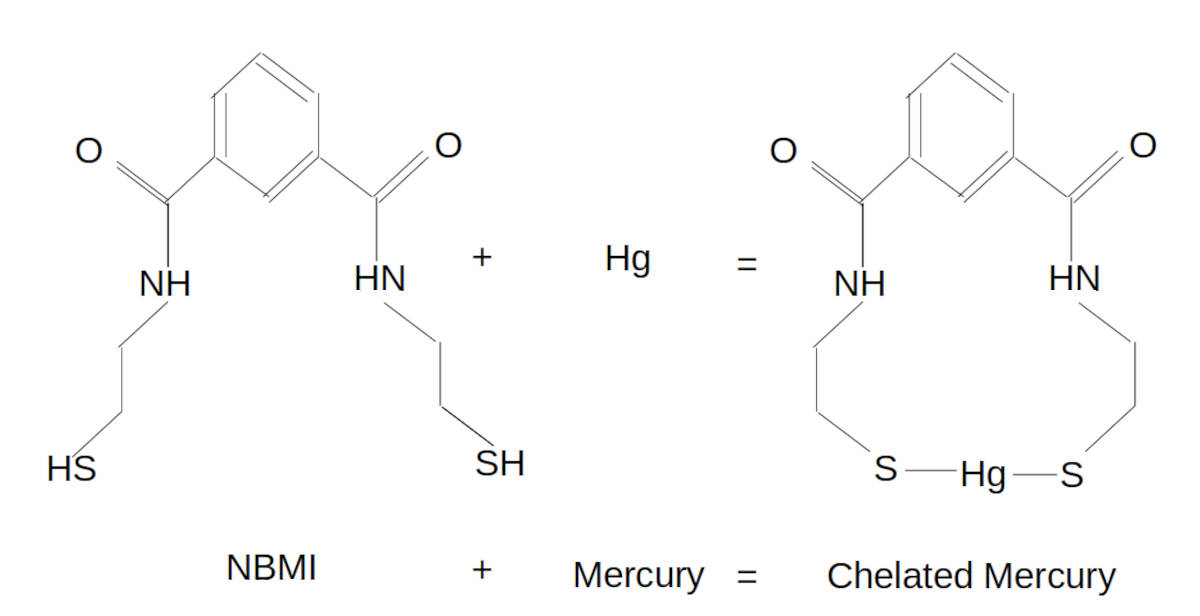
NBMI: as a dithiol chelator it has two sulfur atoms which can bind to mercury with the distinct advantage that the spacing of its sulfur atoms is not fixed and thus can easily accommodate two bonds with a mercury atom with one NBMI molecule unlike ALA, DMPS and DMSA which prefer two molecules to bind to two atoms of mercury.
According to Dr. Boyd Haley PhD, NBMI attaches to mercury to immediately render it non-toxic and requires a temperature of 230 degrees Celsius to break the bond.
NBMI is also a strong anti-oxidant and scavenger of free radicals.
NBMI also has a preference for bonding with mercury.
If NBMI first finds and binds with a free iron atom, NBMI will release that bond in the presence of mercury and form a new and stronger bond with mercury.
It is lipophilic (fat soluble) and can cross the Blood-Brain-Barrier (BBB), cell membranes and enter cell organelles where metal toxicity is most damaging.
Also see this interview with Boyd Haley PhD on NBMI
Ratio NBMI:Hg is 1:1
Animation of NBMI binding to mercury
Glutathione: is the body's natural antioxidant and detoxifier. The strength of the bond with mercury is less than the pharmaceutical chelators above due to it's single sulpher atom. Typically two glutathione molecules are required to bind and chelate a single mercury atom. This molecular representation is of the OPITAC L-Glutathione Reduced tri-peptide composed of glutamic acid, cysteine, and glycine used by Quicksiler Scientific in their "Liposomal Glutathione Complex" product.
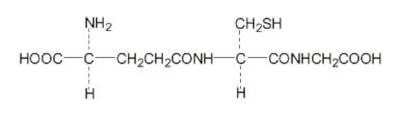
Also see the our PDCure.org project to develope a new mercury chelator.
Challenge Tests:
On their own, blood, urine, stool and hair tests for mercury are poor surrogates to measure the toxins acquired over a lifetime but instead indicate exposures in recent days or months. These tests are only useful as part of a challenge test to see if a chelator is removing mercury and not as a stand-alone test to measure the level of mercury intoxication.
Many medical practitioners will want to perform a "challenge test", also known as a "provocation test" or "mobilization test", as a way to measure the level of mercury in the body and monitor chelation progress. Challenge tests can pull toxic metals even from relatively non-toxic bodies leading to an exagerated positive test result, guiding one to an inappropriate, ineffective therapy and their attendant risk.
The challenge test uses a fixed but large dosage of a chelator to dislodge mercury from the body for measurement with a blood, hair or urine test. Blood tests are transitory and only measure mercury at a given moment in time. This is also true for urine tests. A hair test will measure mercury over a longer but still short period of time. These tests ignore the reality that mercury is not typically mobile and has taken up residence in organs or fat in the body and thus can not be properly measured.
Each chelation agent may preferentially bind to a given metal first, thus hiding the presence of other metals. Mercury can also be tightly bound to body tissue and thus may not be removed until significant amounts of other "preferred" toxic metals have been removed first.
The challenge test just proves the unfortunate truth that our environment, medical community (dental amalgam fillings, thimerosal) and food supply (fish) have intoxicated us with mercury. It does not indicate any comparative level of intoxification although work has begun in this area (ref).
The ACC chelation protocol recommends against this practice as it does not follow their protocol of multiple doses spaced at the half life of the chelation agent so that mercury if dislodged from a chelator, can be picked up by the next round. The American College of Medical Toxicology (ACMT) published a position statement also recommending against the use of this test (ref pdf) as not "accurate or reliable" and is "of no benefit to patient outcome, may actually prove harmful".
The ER guide "Goldfrank's Toxicologies Emergencies" notes that the "provocation test" "tends to increase urinary elimination of mercury, regardless of exposure history and baseline excretion" and states that it is of "dubious value".
Symptoms of mercury toxicity can occur at any blood or urine level. Blood and urine mercury levels reflect recent exposure and do not correlate with total body burden and usually under reflect tissue levels and even more so, CNS levels. Additionally, there are no challenge test protocol standards, laboratory reference ranges or guidance for interpretation of results.
References:- Recommendations for Provoked Challenge Urine Testing (Ruha, 2013)
"... since mercury is ubiquitous in the environment, nearly everyone has at least some low-level exposure to mercury throughout their lifetime, even in the absence of these specific exposures."
"... pre-DMPS urine mercury excretion was associated with the post-DMPS urine mercury excretion in all the groups. It was concluded that the challenge test did not reflect the long-term exposure or body burden"
"DMPS and DMSA challenges produce a rise in urine mercury in all groups of patients, even those without any known exposure to mercury. This is similar to the effect of EDTA, penicillamine, and deferoxamine on other metals in unexposed populations"
"In summary, current evidence does not support the use of DMPS, DMSA, or other chelation challenge tests for the diagnosis of metal toxicity. Since there are no established reference ranges for provoked urine samples in healthy subjects, no reliable evidence to support a diagnostic value for the tests, and potential harm, these tests should not be utilized."
Measuring Mercury Burden:
Measuring one's level of mercury toxicity is difficult as where the mercury ends up, determines its harm to one's body. It is also hard to quantify the level of mercury in one's body. Mercury is bioaccumulative which means that it gets absorbed by body tissue and little is excreated. Blood tests can accurately measure the level of mercury in the blood, but the measurement is of a transient state and has no relevance to the amount of mercury already absorbed by body tissue, nor can it predict how much of the mercury in the blood will be expelled or absorbed. It will merely be a measurement of mercury in the bloodstream and can be a point-in-time indicator of the health of one's current diet or environment. Blood test limits are usually acceptable if less than 10 micro-grams/Liter. A low measurement of mercury in the blood is often mistaken for a low body burden of mercury. This is also true for urine and feces tests as they have the same limitations as blood tests. Hair tests do have the advantage of a measurement over a greater duration of time but otherwise have the same limitations. Hair, urine and feces tests can also be misleading because those that fail to excrete mercury efficiently tend to show low measurements but are more mercury toxic and tend to have a higher body burden of mercury. One can get tested for mercury levels in the blood, hair, urine and feces as well as in body tissue.
Note that organic mercury is excreted through the bile and feces and is not readily detectable in the urine. Urine mercury concentrations are the best indicator of long‐term exposure to elemental mercury while whole blood concentrations are more useful for short‐term, high‐level exposures.
Also see: List of toxin testing companies
References:- Chelation: Harnessing and Enhancing Heavy Metal Detoxification—A Review (Sears, 2013)
"Blood and urine are poor surrogates to measure the toxins accrued over the lifetime (body burden)" - Lessons learned from a fatal case of mercury intoxication (T. Alhamad, 2011)
"Primarily, it is a striking feature of this case that despite treatment with chelators, the patient’s blood mercury level dramatically rose"
Chelation stirs up mercury, raising blood mercury levels, further renewing and complicating the intoxication. - Video of Dr. Boyd Haley on the topic of hair tests and mercury retention
- The Association of Exposure to Lead, Mercury, and Selenium and the Development of Amyotrophic Lateral Sclerosis and the Epigenetic Implications (umich 2010)
DOI: 10.1159/000315405
While not directly about Parkinson's, the paper details examples of why measuring mercury can be so misleading and understate the levels of toxicity.
"There is a large amount of evidence suggesting that mercury is toxic to motor neurons. In animals, mercury accumulates in motor neurons following its injection into blood"
"... the amount of mercury available for excretion may not represent the amount which has accumulated in motor neurons."
- Blood
- Hair
- Stool
- Urine
- Tissue biopsy
- OligoScan
OligoScan: The OligoScan is a toxic metals scan which unlike the blood test can actually measure the mercury burden in tissue which makes the measurement much more relevant.
It uses light spectroscopy (spectrophotometry) to measure the levels directly in the tissue being scanned (typically the hand).
The hand will contain blood, cellular tissue and interstitial space, all of which will contribute to the measurements in the scan.
A measurement of the mercury burden of the hand is expected to be a proxy for the overall body burden.
Measurement is based on the scientific principle that every chemical element absorbs, emits or reflects light (electromagnetic radiation) of a given wavelength.
The more the tissue is concentrated with a given element, the more it absorbs the light of a corresponding specific wavelength (Beer-Lambert's law).
The wavelength of light absorbed is unique for each substance (between 180 and 1000 nanometers).
Measurements of elemental toxins (lead, mercury, arsenic, cadmium, aluminum, ...) is more specific to a given wavelength than measuring organic molecules and compounds.
The amount of absorbance determines the concentration of the substance.
The scale for determining toxicity is a proprietary scale based on European and international standards (WHO, ATSDR).
The Oligoscan is not recognized by the FDA, can not measure toxic metal burden in the brain, central nervous system, kidneys or liver.
Many instruments in the medical, chemical, pharmaceutical and food processing fields use spectrophotometry principals for measurement.
Also see OligoScan
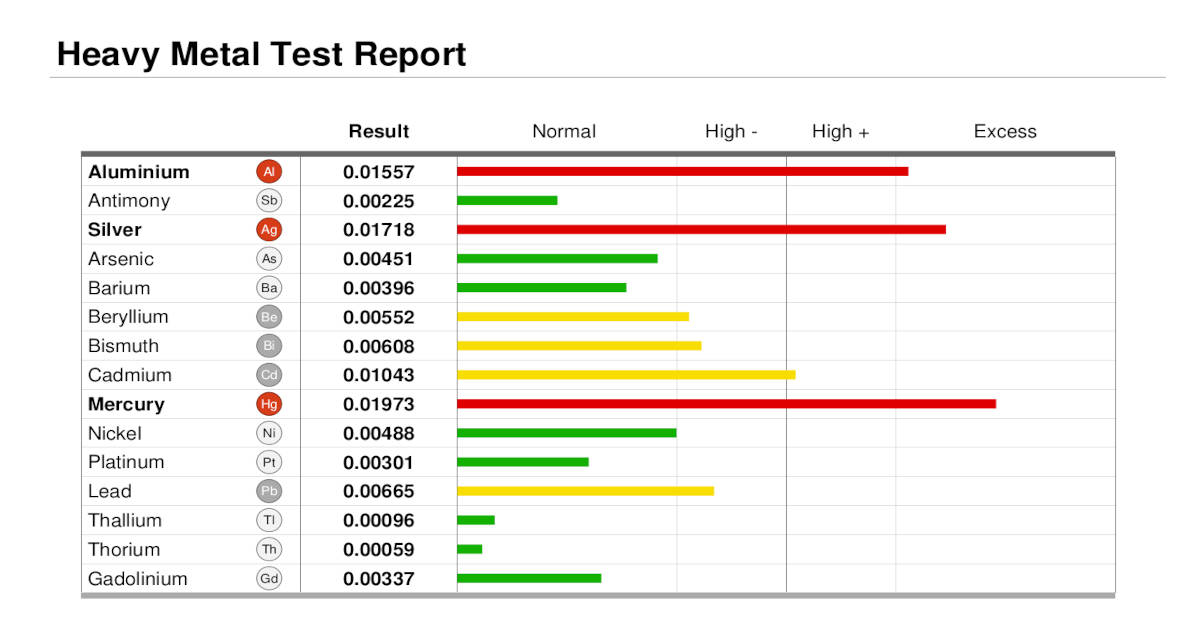
Example OligoScan Heavy Metal Test Report. Note that the result values are not of standard units like micro-grams per liter so comparisons with blood tests are futile. Oligoscans are also correlated to the physiology of an individual and thus comparisons with other patients are not valid. Additional OligoScan tests can be merged into comparison reports to track progress. In this example, silver and mercury are elevated which is a key indication that amalgam dental fillings are shedding material which is being absorbed by the body. Note that early chelation rounds may show an increases in mercury as a result of mercury mobilization when using pharmaceutical chelators like EDTA, DMSA, DMPS or ALA. Also, improper removal of silver-mercury dental amalgam fillings will spike silver and mercury levels.
Opinion: Despite all of its faults and detractions and the fact that no medical governing body approves of its use, it is our opinion that the Oligoscan remains the best tool to measure human body mercury burden.

OligoScan scans four points on the hand.
Operated by Ananda Integrative Medicine
Cyrex: Neurological Autoimmune Reactivity Screen (Array 7): This screen test for toxic heavy metals tests one's immune reaction. Blood test examines the associations between mercury biomarkers and antinuclear antibody (ANA) positivity and concentration. The higher the ANA, the stronger the correlation with mercury levels. An autoimmune antibodies test can detect first signs of clinical toxicity by years compared to other screening tests. Your licensed healthcare professional will perform the blood draw and interpret the results. Regulations governing clinical laboratories prevent laboratory personnel from providing technical assistance to patients.
Predictive Antibodies:- Myelin basic protein IgG + IgA combined
- Asialoganglioside IgG + IgA combined
- Alph and Beta Tubulin IgG + IgA combined
- Cerebellar IgG + IgA combined
- Synapsin IgG + IgA combined

Tri-Test: All mercury is not equal. Elemental inorganic mercury is far less toxic than organic methyl mercury.
This distinction is found in the Quicksilver Scientific mercury "Tri-Test" which measures mercury in the blood, urine and hair and distinguishes the difference in measurements for elemental inorganic mercury (Hg2) and methyl-mercury (MeHg).
Hair tends to sequester and expel methyl-mercury, urine tends to sequester and expel inorganic mercury while blood contains both.
Most traditional tests report "total" mercury rather than individual organic/inorganic mercury levels.
The "Tri-Test" results will also show a plot of urine vs blood levels to show how effective one's body is at expelling mercury.
Also see Quicksilver Metals Testing
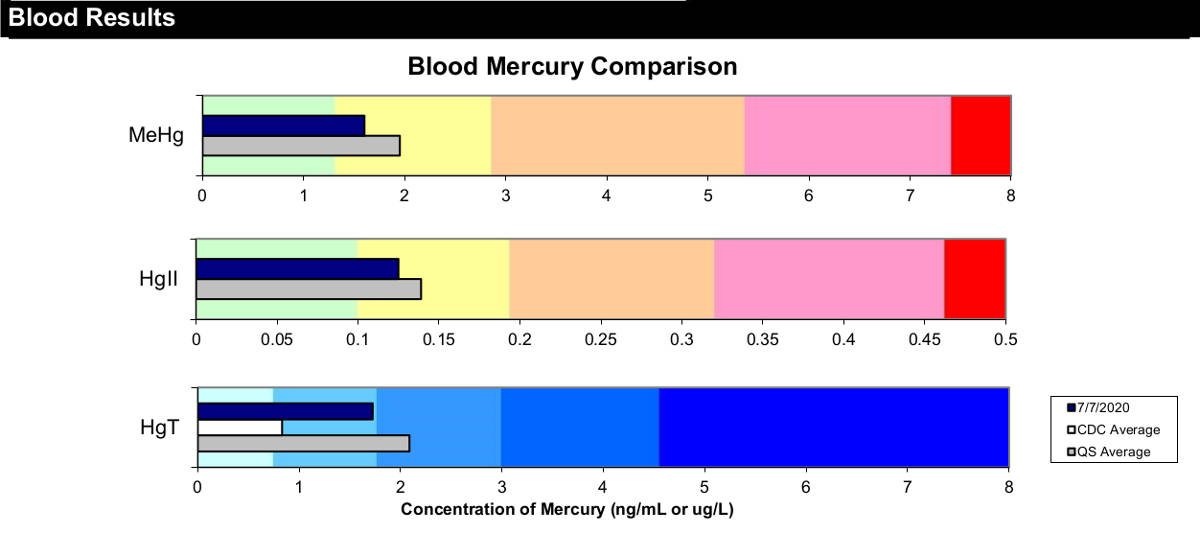
Tri-Test results show the contribution of organic methylated mercury (MeHg) and inorganic mercury (Hgll). This also guides one's choice in chelators as DMSA favors the former while DMPS favors the latter. Clearly in this example, MeHg comprises the bulk of the total mercury burden. The total burden (HgT) is also plotted. Note that hair tests do not account for excretion efficience and can be a false negative as a low measurement can reflect the inability to excrete mercury and may be unable to measure a mercury toxic patient or a high body burden of mercury.

Quicksilver mercury Tri-test: includes two blood vials and urine vial, instructions and mail pouches
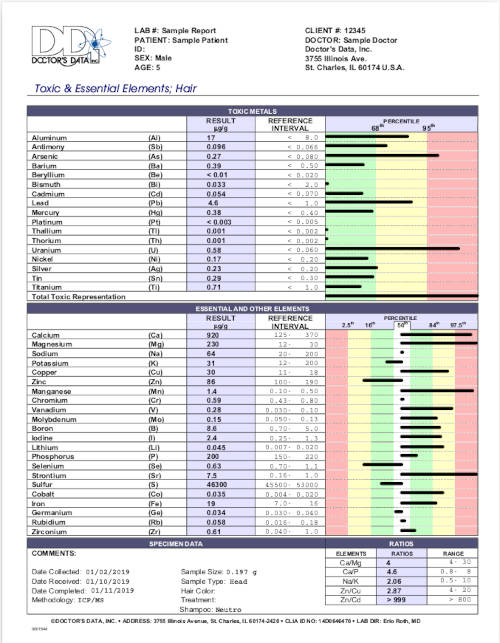
ACC Counting Rules: The Dr. Andy Cutler Chelation protocol "counting rules" applies to hair element analysis results to determine if one is mercury toxic. One can assume that hair tests only measure toxic metals as a momentary status and not the true toxic body burden measured in one's human tissue. The counting rules use the cumulative results from all metals in order to determine the mercury toxicity status. There is no mathematical basis to support this measurement rule hypothesis nor any peer reviewed research. Also hair tests do not measure the body burden of toxic metals stored in a person's organs and tissue but show the available circulating levels at a given point in time.
Counting Rules:- Five or fewer bars from the middle white zone going right
- Five or fewer bars from the middle white zone going left
- Four or more bars in the red zone (either left or right)
- Eleven or fewer in the white and green zone (not touching the yellow zone)
- Missing any two of the above rules by one bar
Meeting a rule equates to being mercury toxic.
This test is applied to a Doctor's Data Toxic and Essential Elements Hair Test (not the Hair Toxic Element Exposure Profile).
Urine Test: The comprehensive toxic metal panel 24 hour urine test is prefered over other urine collection techniques. If using a Quest Diagnostics lab, request test code 37081 which will test for arsenic, cadmium, cobalt, lead, mercury and thalium.
Instructions for the test include abstaining from supplements, vitamins, minerals and non-essential medications. Also avoid shellfish and seafood for 72 hours which will potentially skew the results.
Ignoring the first urination of the morning, collect usine samples over a 24 hour period and return the collection container to the lab.
Organic mercury is not readily detectable in the urine. Urine mercury concentrations are the best indicator of long‐term exposure to elemental mercury.

Quest Diagnostics 24-hour urine test
Stool Test: The GPL Metals Fecal Test shown here tests for metals excreted. One can assume that fecal test only measures toxic metals as a momentary status and not the true toxic body burden measured in one's human tissue. It can show that chelation with ALA or NBMI is effective by showing a pre and post provoking chelation test increase.
It will not show efficacy of chelators like DMSA which excrete mercury via the kidneys.
Note that those who's bodies are not effective excretors of mercury may have a high body burden of mercury but a stool specimen low in mercury.
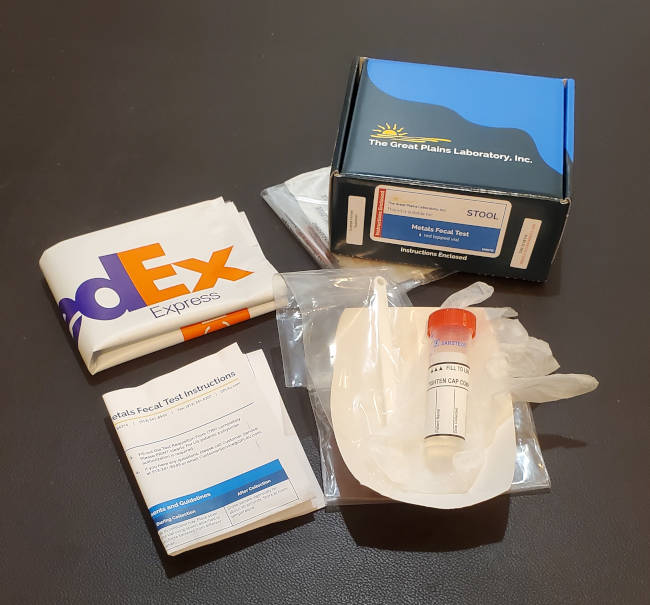
GPL metals feces test kit
Mercury Measurement Limits:
| Measurement | 95th percentile adult | Investigate | Toxic |
|---|---|---|---|
| Blood | 23 nmol/L (4.6 μg/L) | 50 nmol/L (10.0 μg/L) | 200 nmol/L (40.0 μg/L) |
| Urine | 4.0 nmol Hg per mmol creatine | 19.8 nmol Hg per mmol creatine |
Measuring Mercury Conclusion: Mercury will bind with body tissue thus rendering blood, urine, stool and hair tests useless except for measuring recent mercury exposure. A challenge test will almost certainly render a positive result as a lifetime of bioaccumulative exposure will result in the mobilization of mercury. The challenge test can only conclude that one possesses a mercury burden but does not offer a comparative measurement. The challenge test may be more realistic in that it often concludes that a mercury burden exists while a stand-alone blood, urine, stool or hair test will suppot a sense of denile that no level of mercury intoxification exists at all.
There is no good way to measure mercury toxicity. What really matters is a measurement of the brain burden of mercury and only an autopsy can measure this accuratly.
Vendors of Chelation Agents:
The following is a table of chelation agent vendors.
| Vendor | DMSA | DMPS | ALA | RLA | Notes |
|---|---|---|---|---|---|
| Living Supplements | 0.25, 1, 3, 4, 6.25, 12.5, 20, 25, 50, 100 mg | 0.25, 1, 2.5, 5, 10, 15, 25 mg | 0.25, 0.5, 1, 3, 5, 6.25, 12.5, 18.75, 25, 33, 50, 75, 100 mg | no | DMSA and DMPS capsules includes 50 mg vitamin C unless otherwise noted. They also have an ALA / DMSA / vitamin C combo capsule (12.5 or 25 mg of both ALA and DMSA). Shipped from UK. Located in South Africa (may have to inform your credit card company to turn off fraud blocking for a purchase) |
| MandiMart UK | yes | no | yes | no | UK reseller of Living Supplements DMSA and ALA. |
| Quicksilver Scientific [alt] (Quicksilver Canada) | no | no | no | 48 mg | Detox Cube bundle which includes Glutathione, "Liver Sauce" (a proprietary mix of ingredients including RLA) and GI binders (IMD: Intestinal Metals Detox). |
| Dr. McGuire's dental wellness website | no | no | 150 mg | no | They also have vitamins and supplements for detox: Selenium, free form amino acids, vitamin C, multi-vitamin, multi-mineral |
| Everything Spectrum | no | no | 5, 12, 25 mg | no | They also have "Essential 4" vitamins (C:750mg,E:400IU,Mg:300mg,Zi 30mg), adrenal cortex (250 mg) and enzyme supplements |
| Stop Aging Now (Vitamin Research Products) | no | no | 100 mg | no | Sold as a capsule which bundles ALA with B12, DHA, curcumin and bacopa extract and is sold as "Brain Energizer Complex" |
| Thorne Labs | no | no | 300 mg | 100 mg | They also have a heavy metal detox bundle |
| Woodland Hills Pharmacy | custom orders. 12.5 - 500 mg | no | yes | no | Compounding pharmacy fills to order for specified dose and quantity. Can combine DMSA with ALA. Prescription required. |
| CareFirstRx | custom orders. Any mg | no | no | no | Compounding pharmacy fills to order for specified dose and quantity. Prescription required. |
| NewRootsHerbal | no | no | 125, 250 mg | no | ALA only |
| NutraBio | no | no | 300 mg | no | ALA only |
| NuVision | 100 mg | no | yes | no | DMSA includes 200 mg of vitamin C and 50 mg glycine. ALA is blended with other compounds as a brain supplement. |
| Vibrant Life | no | no | 100 mg | no | Also available: Liposomal EDTA with R-Lipoic Acid and Glutathione/EDTA combination |
| Supersmart DMSA | 400 mg | no | no | no | High dosage |
Note that DMSA and ALA are often sold in doses of 100mg or more. This is a high dosage and not recommended for initial rounds or ever. Dosages of this magnitude for a mercury toxic person could have very harmful consequences.

DMSA

DMPS

ALA
One can also do an internet search for local "compounding pharmacies" in your area who can generate the dosage and quantities of a chelation agent custom made for you. Another alternative is to buy chelation compounds in bulk and make your own capsules. Customization allows mixing DMSA with ALA and essential 4 core vitamins all in one simple capsule at your own preferred dosage.
Bulk powder: Capsules and filling trays:
Jewelry scale to measure milligrams of bulk powder
NBMI: the chelator NBMI is only available in bulk powder form and can be measured (spoon) or weighed to achieve the desired dosage.
Available from:- EmeraMed sourced from PCI (USA) expected availability in 2024 (estimate) after FDA approval or sign up for their "Early Access Program".
- MedChemExpress (USA) for laboratory research use
- Aobious (USA) for laboratory research use
- TRC: Toronto Research Chemicals (Canadian) for laboratory research use
- Fandachem: 351994-94-0 (off brand China source) - two versions of NBMI are sold, the preferred version is synthesized with a Class 3 solvent Ethyl Alcohol (EA) rather than the less favored dichloromethane (DCM) (contact Ren Dylan).
- Medkoo (China with an office in North Carolina) synthesized with solvent dimethylforamide (DMF)
- Octagon Chemical (China)
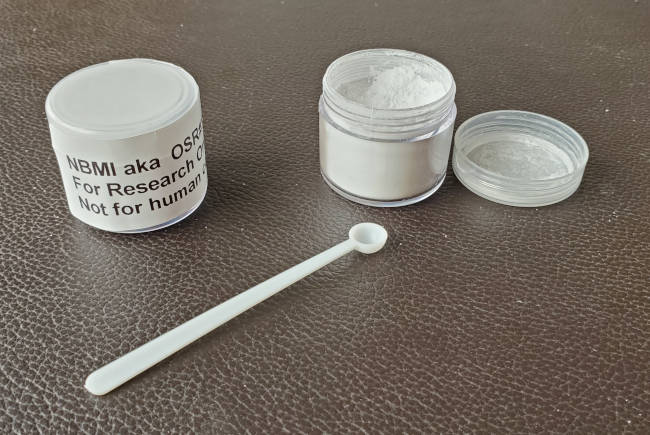
NBMI bulk powder
Compound Testing Laboratories:
The following is a list of testing laboratories which can verify the purity of chelation compound samples sent to them.
- Spectrix Analytical Services - labs in North Haven CT and Oak Park IL
Chelation Detox Protocols:
Warning: All chelators and chelation protocols are considered high risk for permanent brain, kidney and liver damage. Chelators will often bond to mercury in body tissue and drop it in high mercury affinity areas like the brain and central nervous system. This relocation can cause extreme damage as the brain and central nervous system are very sensitive to the toxic effects of mercury. Continued efforts to chelate often makes the condition much worse leading to extreme mental anguish and suicidal tendencies.
None of the chelation protocols below have any extensive published or scientific studies showing efficacy. This website does not endorse chelation or any particular chelation therapy due to the dangers and extreme risks involved.
The safest action is to remove the source of mercury exposure (eg. removal of dental amalgam dental fillings using IAOMT SMART protocols and cease seafood consumption) and avoid the consumption of any substance (chelator, or high thiol food or supplement) which can mobilize mercury. One must recognize that natural, unmodified excretion pathways including employing a sauna to provoke sweat, is the best course of action.
Dr Chris Shade's Mercury Detox Protocol:
This protocol relies on glutathione to transport mercury out of body tissue, organs, the brain and central nervous system and is unique in that it also supports the evacuation by the liver and GI tract. Chris Shade's protocol also relies on unique products he has developed and sells through the company he founded, Quicksilver Scientific. Quicksilver products can be purchased individually or as bundles. Products which support their mercury detox protocol are based on their Liposomal Glutathione Complex for mobilization, Liver Sauce to support liver evacuation of toxins and their Intestinal Metals Detox (IMD) binder which is specifically tailored to bind to mercury and other heavy metals and escort them through the GI tract without re-absorption. The mercury detox products can be purchased individually or as a "Detox Cube" bundle with additional vitamin and nutrient support and additional cost. Liver Sauce can be purchased individually or bundled with their Ultra Binder for additional GI support. This Liver Sauce and Ultra Binder bundle is sold as the "PuchCatch Liver Detox" product. Note that their EDTA products are for lead and cadmium chelation and not for mercury detox.
The PushCatch Liver Detox product is for addressing general detoxification and can be extended to include mercury detoxification by adding their Liposomal Glutathione Complex product. Basic PushCatch "Liver Sauce" uses a combination of R-Lipoic Acid (RLA: nature's version of ALA), glutathione production and the enzymes that work with it. Dr. Shade feels that reliance on glutathione avoids the half life problem of pharmaceutical chelators as the body regulates the levels in the bloodstream which never go to zero. The focus of this protocol is on liver detoxification and balancing inflammatory pathways. The "Liver Sauce" includes a multitude of botanicals (myrrh, dandelion, gentian, goldenrod), liver treatments (nano milk thistle, DIM, quercetin, luteolin, r-lipoic acid), binders (activated charcoal, bentonite clay, chitosan, IMD) and GI lining stabilizers (Fibregum Bio, acacia gum and BiAloe). Dr. Shade's position is that RLA is an NRF-2 (Nuclear factor erythroid-related factor 2, cellular detox gene) up-regulator (rather than a chelator) which makes the body produce more glutathione which leads the body to detox in a “natural” way. Dr. Shade also sees R-LA as an AMPK (promotes ketosis: cardio and metabolic health) up-regulator as well as a PGC-1-alpha (mitochondria biogenesis) up-regulator. He also believes that R-LA has a 20 fold better absorption than ALA (ref). This protocol encourages the liver to metabolize toxins to dump them with bile into the GI tract where the toxins cling to binders so that they will be evacuated and not re-absorbed.
We feel an obligation to point out that while these Dr. Shade Quicksilver detox products might be sold for detoxifying the body of mercury, there is no statement on the Quicksilver Scientific website or in their product literature that makes any such claim. There have also been no medical studies published on the effectiveness of their products to remove mercury.
Three part protocol:- Glutathione: (mobilize and neuroprotective)
- Liposomal transport system employed so that glutathione (GSH) can cross membranes. Liposomal encapsulation is a pharmaceutical process to encapsulate (typically in lecithin) the medicine to be deliverd so that it can transport through tissue membranes or be protected from digestion and be absorbed.
- Liposomal-Glutathione Provides Maintenance of Intracellular Glutathione and Neuroprotection in Mesencephalic Neuronal Cells (Zeevalk, Bernard, Guilford, 2010) PDF
"In Parkinson’s disease (PD), there is a 40–50% decrease in total GSH. The decrease in GSH is found in the brain region most affected in the disease, i.e., the substantia nigra and is thought to be one of the earliest dysfunctions in the disease process."
"Exogenous Liposomal-GSH is More Efficacious than Non-Liposomal GSH for Replenishment of Intracellular Glutathione Levels"
"... liposomal-GSH was 100-fold more potent in serving as a source for intracellular GSH repletion."
"These findings provide evidence for utilization of liposomal-GSH via the endosomal/lysosomal pathway." - autophagy and lysosomes dysfunction are a root cause of alpha-synuclein aggregation and Parkinson's pathogenesis. In this study, glutathione was found to be a crutial ingredient for proper lysosomal function (protein recycling).
"Two major findings result from these studies: firstly that liposomal-GSH can be utilized for repletion and maintenance of intracellular GSH in neuronal cells and secondly, that liposomal-GSH can provide significant protection to neurons in a model system relevant to Parkinson’s disease."
"elevation of extracellular GSH may pose potential toxicity problems that increase neuronal vulnerability during ischemia or enhance toxicity involving NMDA receptors. ... Encapsulation of GSH into lipid vesicles may avoid the potential toxicity to neurons associated with extracellular GSH elevation and may facilitate drug delivery to cells as has been shown for other liposomal preparations."
DOI: 10.1007/s11064-010-0217-0
- Liver Sauce ingredients: (push)
- quercetin: flavonoid to modulate pro-inflammatory mast cell activity. Ubiquitously contained within natural plants such as green tea, and vegetables.
Also see: - luteolin: flavonoid to modulate pro-inflammatory mast cell activity. Luteolin is found in abundance in celery, green pepper, and dandelions.
Also see:- Anti-Oxidant, Anti-Inflammatory and Anti-Allergic Activities of Luteolin (2008)
- Luteolin suppresses inflammation-associated gene expression by blocking NF-κB and AP-1 activation pathway in mouse alveolar macrophages (2007)
- Luteolin and luteolin-7-O-glucoside inhibit lipopolysaccharide-induced inflammatory responses through modulation of NF-κB/AP-1/PI3K-Akt signaling cascades in RAW 264.7 cells (2013)
- DIM (diindolylmethane): hormone regulator. Promotion T-regulatory dominance (immune moderating). Stimulates Nrf2 antioxidant signal.
Also see:- 3,3′-Diindolylmethane Stimulates Murine Immune Function In Vitro and In Vivo (2008)
"Oral administration of DIM markedly increased the concentration of 4 out of 22 serum cytokines tested over this time period, ..."
"DIM’s capacity to stimulate proliferation in naïve splenocytes suggests an inherent potential for this compound to stimulate immune responses. T cell activation can result in cytokine production, cytokine receptor expression and finally proliferation of the activated T cells"
"Cytokines promoting the development of T-cell-mediated immunity can induce or enhance the antitumor and antimicrobial immunity"
"DIM is a potent stimulator of immune function and can directly affect splenocyte and macrophage function. These properties might contribute to its well-documented antitumor and antiviral effects." - Low concentrations of diindolylmethane, a metabolite of indole-3-carbinol, protects against oxidative stress in a BRCA1-dependent manner (2009)
- Epigenetic Modifications of Nrf2 by 3,3′-diindolylmethane In Vitro in TRAMP C1 Cell Line and In Vivo TRAMP Prostate Tumors (2013)
- 3,3′-Diindolylmethane Stimulates Murine Immune Function In Vitro and In Vivo (2008)
- R-Lipoic Acid: Nrf2 up-regulator used to drive an increase production of glutathione which is the body's natural detox excretion pathway.
Also see:- Regeneration of glutathione by α-lipoic acid via Nrf2/ARE signaling pathway alleviates cadmium-induced HepG2 cell toxicity (2017)
DOI: 10.1016/j.etap.2017.02.022 - Protective effect of lipoic acid against oxidative stress is mediated by Keap1/Nrf2-dependent heme oxygenase-1 induction in the RGC-5 cellline (2012)
"Oxidative stress plays a key role in neurodegeneration of CNS neurons such as in Alzheimer disease, Parkinson's disease and glaucoma. R-α-lipoic acid (R-LA) has been shown to have a neuroprotective effect through its antioxidant activity"
DOI: 10.1016/j.brainres.2012.12.041 - Effect of Nrf2 activators on release of glutathione, cysteinylglycine and homocysteine by human U373 astroglial cells (2013)
- Regeneration of glutathione by α-lipoic acid via Nrf2/ARE signaling pathway alleviates cadmium-induced HepG2 cell toxicity (2017)
- milk thistle: liver protection and bile flow stabilization
Also see:- Hepatoprotective effect of silymarin (2014)
"There is substantial evidence suggesting that silymarin treatment improves hepatic diseases. However, some of the data are contradictory" - Milk Thistle: Effects on Liver Disease and Cirrhosis and Clinical Adverse Effects (2000)
"Clinical efficacy of milk thistle is not clearly established"
- Hepatoprotective effect of silymarin (2014)
- quercetin: flavonoid to modulate pro-inflammatory mast cell activity. Ubiquitously contained within natural plants such as green tea, and vegetables.
- IMD GI tract binder:
- Thiol-functionalized silica has an affinity for mercury and prevents mercury from being re-absorbed. IMD contains purified silica with covalently attached thiolic (sulfur) metal-binding groups. IMD does not enter the bloodstream, and thus will not lead to the mobilization of mercury but will escort mercury ouot of the body via the inttestine. IMD is typically taken on an empty stomach.
- Thiol-Functionalized Mesoporous Silica for Effective Trap of Mercury in Rats J of Nanomaterials (Wang et al, 2016)
Conclusion: "Hence, thiol-functionalized mesoporous silica has potential application in preventing and treating heavy metal poisoning."
The weekly protocol is maintained for five days of product consumption followed by two days off for the body to recover and then repeated. Protocols can be custom formulated by a Quicksilver Scientific consultant (Dale White).
- Week 1:
- Liposomal Glutathione Complex: 1/2 tsp/twice a day
- Liver Sauce: 1 tsp/twice a day
- IMD taken with vitamin C and water 30 minutes after Liposomal Glutathione Complex and Liver Sauce: 1 scoop/twice a day
- Week 2:
- Liposomal Glutathione Complex: 1 tsp/twice a day
- Liver Sauce: 1.5 tsp/twice a day
- IMD taken with vitamin C and water 30 minutes after Liposomal Glutathione Complex and Liver Sauce: 2 scoops/twice a day
- Week 3-8:
- Liposomal Glutathione Complex: 1.5 tsp/twice a day
- Liver Sauce: 2 tsp/twice a day
- IMD taken with vitamin C and water 30 minutes after Liposomal Glutathione Complex and Liver Sauce: 3 scoops/twice a day
- Week 9+:
- Liposomal Glutathione Complex: 1 tsp/twice a day
- Liver Sauce: 2 tsp/twice a day
- IMD taken with vitamin C and water 30 minutes after Liposomal Glutathione Complex and Liver Sauce: 3 scoops/twice a day
No food should be consumed for 30 minutes after taking IMD as it will help expel the food and its nutrients. Quicksilver Scientific's Ultra Binder can be taken with IMD to help flush additional toxins. Quicksilver Scientific's Quinto Isotonic mineral supplement can be added 10 minutes before meals or on an empty stomach (twice daily fo 4 months, twice weekly thereafter).
Also see:- QuickSilverScientific: mercury detox
Emerson Ecologics (alternate)
(Quicksilver Canada) - QuickSilver Scientific Facebook Group

Quicksilver: Mercury detox
- Liposomal Glutathione Complex (mobilization and neuroprotective)
- LiverSauce (push)
- Intestinal Metals Detox (IMD) (catch: GI binder)

Quicksilver: "Push-Catch" liver detox
- LiverSauce (push)
- UltraBinder (catch: GI binder)
Dr. Boyd Haley PhD Mercury Chelation Protocol:
Boyd Haley has a PhD in biochemistry, was an NIH funded researcher at the University of Wyoming and at the University of Kentucky (and professor of chemistry 1996-2005) and is the principal developer of NBMI as a chelator for mercury detoxification and founder of EmeraMed, a biotech firm created with the goal of getting NBMI FDA approved as a treatment for mercury toxicity in both humans and animals. Dr. Haley's protocol is the latest of the mercury chelation protocols and employs a new chelator, NBMI, claimed by its producer, EmeraMed, to not have the mercury mobilization problems and the side effects of some of the more established chelators like DMSA, DMPS, ALA and EDTA. The protocol recommends starting with low doses (25 mg a day) and working on up to 300 mg (or 4 mg/kg of body weight ref) a day. Test data has shown that low doses (100 mg) of NBMI get consumed by the body while scavenging free radicals and it is at the higher doses (300 mg) that noticable mercury chelation occurs (ref). Doses can be administered 1 to 4 times a day as it has an 18+ hour half-life allowing infrequent doses. NBMI can be added to fat (olive oil or corn oil or butter, etc.) to improve absorption. One may even begin using NBMI transdermally by mixing it with olive oil and applying it to one's skin.
Experimental use of NBMI includes a Swiss clinic which has treated a bed bound patient with atypical Parkinson's, enabling the resumption of use of their right leg and hand. The assumption is that their root cause was mercury toxicity and that NBMI detoxified the patient (ref).
Mercury toxic people must start slow with low doses to avoid overwhelming the body. When mercury bonds with NBMI it renders mercury non-toxic and makes an almost unbreakable bond thus it avoids the problems of mobilization and re-absorption common with other chelators. NBMI still possesses its own risks including high doses which may harm the brain and organ tissues by detaching mercury from cell protein and inducing an inflammation response to signal cellular repair (Ref. Facebook Mercury chelation group file: OSR-Emeramide-NMBI-Irminix.pdf by Aron). Symptoms are reported to include a knotted tension feeling as the NBMI bonds and removes mercury. Chelation is never risk free. Note that NBMI is not FDA approved and there are no independent medical peer reviewed studies on its effectiveness as a chelator to lower the body burden of mercury.
NBMI is not known to chelate essential minerals except zinc and copper which can be supplemented three hours before or after taking NBMI (ref).
Note that the P450 enzymes ultimately break down NBMI and NBMI will not remove mercury from the body.
Also see:- IAOMT Presentation on NBMI by Boyd Haley PhD, developer of NBMI (2016)
- EmeraMed.com: Emeramide
- Mercury chelation Facebook group - questions and answers, discussions and advice. Common theme: like other chelation groups, starting with high doses leads to feelings of misery. Lots of testimonies from those who were on the ACC protocol, felt miserable, then tried NBMI and felt better.
- Irminix / NBMI / Emeramide / OSR Facebook Group - Facebook group gets archived/locked on the weekends to give admins the weekends off
- Amelioration of Acute Mercury Toxicity by a Novel, Non-Toxic Lipid Soluble Chelator NBMI: Effect on Animal Survival, Health, Mercury Excretion and Organ Accumulation - 2012 Clarke, Haley et al
DOI: 10.1080/02772248.2012.657199
"The most significant finding was that NBMI treatment did not reduce the mercury levels in the organs of rats treated with NBMI." - it doesn't work
"NBMI treatment administered to rats acutely exposed to toxic levels of Hg did not significantly alter Hg excretion in urine or feces"
Dr. Boyd Haley PhD
Dr. Andy Cutler PhD Chelation (ACC) Protocol:
The ACC protocol addresses the chelation of mercury and the removal from tissue, organs and from the brain. The details of the ACC protocol are covered in the book "The Mercury Detoxification Manual". The ACC protocol can be performed by one's self or by a medical professional. This protocol involves the use of the chelation agents DMSA or DMPS and ALA taken orally in a given dosage and schedule, starting with low "microdoses" and working up to higher doses over time.. The scheduled times are during a full 24 hour day and not just a daytime schedule. The schedule is dependent on the half life of the chelation agents to maintain an active level of continuous chelation and mercury removal and missing a dose by an hour or more will require a round to be stopped. The ACC protocol takes a commitment and discipline over the course of a few years. The ACC protocol also has the significant risk of inducing further damage from mercury toxicity because of the use of DMSA or DMPS chelators which are known to redistribute mercury to the brain and nervous system.
Stage 1 - Body: The ACC protocol begins with a general removal of mercury from the body in at least 3 or 4 rounds (one round = three 24 hour "on" days separated by a four to eleven day "off" period) using DMSA.
The schedule for each round is 25 mg as stated on page 65 of the manual or 12.5 mg as stated on the ACC website (editor's note: these are conflicting but high dosages. After a round, patients often recommend a light dosage start at 1 mg or 3 mg) of DMSA every 4 hours (for example: 7am, 11am, 3pm, 7pm, 11pm and 3am) for three days.
The dosage can be adjusted higher or lower depending on tolerance.
Start with very low dosages so as to not stir up and redistribute too much mercury.
The chelators DMSA and DMPS will mobilize and redistribute mercury, so the dosage should be chosen to minimize this effect.
Jumping to much higher dosages by more than 50% are NOT encouraged. Tolerated low dosages are best.
Fatigue, kidney irritation and symptoms of mercury toxicity are the most common side effects.
Repeat the 3 or 4 rounds at higher dosages until you are using a 25 mg dosage of DMSA with few side effects.
The dosage should remain steady for the 3 or 4 rounds before an increase is introduced.
These initial rounds are intended to lower your body burden of mercury and create a mercury concentration gradient which favors the flow of mercury from the brain to the body rather than vice-versa.
The protocol recommends that the patient "Keep the dose low and go slow".
While the duration is not specifically stated, it may take many months to achieve your goals.
Warning: research has shown that the use of DMSA or DMPS alone will migrate mercury from the body to the brain and central-nervous-system (CNS)
(for more on this see "Dangers of Chelation Therapy" below).
This is amplified by the fact that neither DMSA, nor DMPS can cross the blood-brain-barrier (BBB).
It has been speculated that DMSA and DMPS should never be used without an additional chelator which can cross the BBB (eg ALA) but there is no research to verify this notion.
Stage 2 - Body and Brain: Once you have reached the 25 mg dosage of DMSA, the ACC protocol recomends taking 12.5 mg ALA and 25mg DMSA every 3 hours for three days "on" followed by a four to eleven day "off" period of no chelation agents. DMSA is used in stage 1 to reduce your body burden of mercury before you start taking mercury out the brain and internal organs with ALA in stage 2. If ALA is not tolerated well, drop the dosage to as low as 3mg. The protocol details schedule changes to accommodate ALA intolerance and getting back on schedule. Note that ALA should not be taken for more than 3 consecutive days. The protocol (30mg DMSA and 12.5mg ALA, or 25mg DMSA and 17.5mg ALA - ref: ACC website) is followed until the target dosage of DMSA and ALA are reached (200 mg ALA/dose for 6 months - ref: ACC website and Detox Manual pg 71). Final dosage of ALA can be more accurately calculated as 3 mg for each kg of personal weight.
The body does not store or release mercury in a consistent fashion, which affects chelation. This manifests itself as inconsistent results and problems round to round. For this reason it is important to find a low and tolerable dosage to repeat for a long duration rather than constantly trying to raise the dosage to optimize progress.
Reintroduction of essential minerals (magnesium: 100-200 mg with meals, zinc: 50 mg/day), vitamins (C and E), essential fatty acids (omega 3) and Adrenal Cortical Extract (ACE) supplement is required to counter the adverse effects of mercury and mercury chelation. The ACC protocol has a schedule and dosage for supplements. One performs a urine or hair sample test for mercury to monitor dropping levels (hopefully). The chelation regiment is repeated until the mercury drops below an acceptably low level. Note that this is purely an indication of mercury levels as true levels in organs and the brain can not be tested. The ACC chelation protocol can last 1 to 4 years.
While this chelation protocol does not have any medical peer reviewed studies as to its effectiveness, there are plenty of medical studies on the effectiveness of the DMSA and DMPS as chelation agents for the removal of mercury. There are also studies which show how these same chelators will mobilize mercury and will make the consequences of mercury toxicity worse.
Also see:- The Dr. Andy Cutler PhD chelation (ACC) Protocol (DMSA or DMPS and ALA)
- ACC mercury and heavy metal detox Facebook group - questions and answers, discussions and advice. A lot of mercury toxic people vocalizing their misery due to mercury mobilization and redistribution from taking DMSA, DMPS or ALA. Administered by a very intolerant group which does not allow for the exchange of ideas or updates to old methodologies nor the mention of, or comparison to, other protocols. They ignore medical texts, new research and medical technology and take down posts that mention unfavorable views about the ACC protocol, and will delete you from the group. Posting facts which are not included in the "Mercury Detoxification Manual" or do not follow the most literal adherence to the ACC protocol are also deleted and will get you expelled.
- Vitamin C, Glutathione, or Lipoic Acid Did Not Decrease Brain or Kidney Mercury in Rats Exposed to Mercury Vapor - ACC chelators don't work (Aposhian et al, 2003)
DOI: 10.1081/clt-120022000
"The purpose of this study was to test the hypothesis that DMPS, DMSA, GSH, vitamin C, or lipoic acid, alone or in combination, would decrease brain mercury in rats exposed to elemental mercury vapor. Our results clearly indicated that none of these agents did so."
"Insulin has been claimed to increase the uptake of drugs across the blood-brain barrier. If it did this for DMSA, brain Hg might be decreased. It was not."
"Alpha-lipoic acid is also being used in combination with DMSA or DMPS for treating autistic children. It was not effective in decreasing brain or kidney Hg in our studies." - A case for microdosing the chelator: if the mercury levels exceed the lowest-observed-adverse-effect levels (LOAELs), a cellular absorption cascade of mercury can follow.
Microdosages of the chelator keep the mercury redistribution levels low thus keeping mercury toxicity effects low.
- The Toxicology of Mercury and Its Chemical Compounds. Critical Reviews in Toxicology (Clarkson, Magos, 2006)
"Clearly some minimal threshold dose of methylmercury is needed to elicit a toxic effect. It appears that once this threshold dose is exceeded, a process is triggered that takes months to finally result in an abrupt appearance of signs and symptoms of severe poisoning."
- The Toxicology of Mercury and Its Chemical Compounds. Critical Reviews in Toxicology (Clarkson, Magos, 2006)
- Example Schedules:

Authors: Dr. Andrew Hall Cutler PhD, PE and Rebecca Rust Lee
ISBN 978-0-9676168-4-1
Dr Tom McGuire's Mercury Detox Process:
This author wrote his book to de-mystify the process of mercury detoxification. He supports the body's ability to remove mercury in a process that is based on the premise of starting low and go slow. The book is for those who are about to have their dental amalgam fillings removed and want to follow-up and remove the accumulated mercury from their body. It covers all of the nutrients, vitamins, and minerals needed for your body to manufacture enough antioxidants, and other substances, necessary to rebuild and maintain its defense system against the continuous onslaught of mercury, toxins, oxidation, and toxic metals. The book also covers supplements that aren’t considered vitamins or minerals but are essential to overall health and provide the nutritional support for a mercury detoxification program. It dedicates a chapter to monitoring your progress, chelators and oral health. Note that the choice of chelators used by this protocol (cilantro, chlorella, DMSA, EDTA, ALA, RLA) can and will mobilize mercury causing further toxicity and damage.
Tom is also the author of "The Poison in Your Teeth" and "Healthy Teeth - Healthy Body".
Also see:- DentalWellness4u.com
- Mercury Detoxification - PDF version of the book

Author: Dr. Tom McGuire DDS
Goldfrank's Toxocologic Emergencies:
This is the toxicology textbook you will find in hospital Emergency Rooms and poison centers. The book follows the FDA advisement that dental amalgam exposure is safe and does not lead to poisoning. It does give an exemption for rare cases of immunologic hypersensitivity.
The book does cover mercury chelation with Dimercaprol (BAL) and Succimer (DMSA) and advises for acute and chronic poisoning.
- Acute poisoning: occurs if one is exposed to an elevated dose that sends the body into an immediate toxic reaction. Symptoms typically appear within minutes and can deteriorate into a life-threatening situation.
- Chronic poisoning: slow accumulation in the body over time, slowly damaging organs and tissue causing insidious side effects and poor health.
Dimercaprol (BAL): administer for 10 days in decreasing dosages of 5 mg/kg/dose every 4 hrs for 48 hrs, then 2.5 mg every 6 hrs for 48 hrs followed by 12 hr dosages for a week. This regiment was derived from their chelation of lead protocol and is for acute inorganic mercury poisoning. The book noted that BAL should not be used for organic mercury poisoning as it is more likely to do harm by mobilizing mercury to the brain.
Succimer (DMSA): 10 mg/kg orally 3 times a day for 5 days, then twice a day for 14 days if the GI tract is clear. The book recommends Succimer for those who are not acutely ill or who have been chronically poisoned by inorganic mercury. When chelating methy-lmercury (organic mercury), DMSA is credited with a decrease in brain and body mercury which is in contrast with ACC which limits recognized effectivity to reducing body burden only. Note that this guide recommends an extremely high chelator dosage when scaled for body weight, much higher that other protocols like ACC. High initial doses present their own dangers and should be viewed with caution.
The book acknowledges the use of DMPS and D-Penicillamine but states that BAL and DMSA are the treatments of choice and that DMSA is preferred for those who are not acutely ill. Note that their choice of chelators used will mobilize mercury causing further toxicity and damage.

Authors" Nelson, Hoffman, et al. 2019
Other Protocols:
Other protocols exist but seem incomplete.
- Dr. Axe: Detox with Chelation Therapy - EDTA based chelation
- Chelation Therapy using EDTA either through direct intravenous injection or by oral ingestion in pill form
Physician Association Protocols:
Physician chelation training and certification.
- International College of Integrative Medicine
- ACAM: American College for Advancement in Medicine - Chelation certification
- Advanced Medical Education and Services Physician Association
- Autism Research Institute
Trial to Assess Chelation Therapy (TACT):
Trial to Assess Chelation Therapy (TACT) was a chelation study sponsored by the US National Institutes of Health (NIH) using disodium ETDTA as the chelation agent to test the safety and efficacy of chelation therapy. Primarily the study determined that it benefited those with coronary heart disease with an 18% reduced risk of a cardiac event and that vitamin and mineral supplements had little effect on the outcome. There were two studies performed, TACT and TACT2. The TACT study found EDTA (NaEDTA specifically) to be a safe chelation agent for adults. The TACT study did not address mercury or Parkinson's disease.
Also see:
Comparison of Chelation Therapy Protocols:
The various chelation therapy protocols are often in conflict and do not define a consensus leading to patient confusion on such an important issue. Sorry. The patient must educate themselves and chose a path. Local holistic practitioners are often not of much help either as it is easy to educate oneself to a level that only stumps the practitioner when approached with in depth questions. Choose a practitioner who is familiar with the various protocols and not just with the directions on the side of the chelation agent container.
| Chealation Agent or Supplement | Dr Shade | Dr Cutler (ACC) | Dr McGuire | Dr Haley | Notes |
|---|---|---|---|---|---|
| Cilantro | not mentioned | never | light dose with DMSA | no | Half life unknown. Dosage unknown. Also known as coriander leaves, dhania or Chinese parsley. |
| Chlorella | not mentioned | never | yes | no | ACC warns that it may cause mercury redistribution. Often used as a GI tract absorbant to help excrete mercury mobilized by a chelation agent. |
| Dandelion | yes | no, hard on the liver | ?? | no | Contains luteolin, a flavonoid known to inhibit inflammation |
| DMSA | not mentioned | yes | yes, but optional | no | ACC protocol has dosage taken at half life to always have chelation agent available to pick up droped mercury. Other protocols don't cover this approach. DMSA does not pass through the Blood-Brain-Barrier (BBB) or through cell walls and must work with other chelators that do, to be completely effective. |
| EDTA | primarily for lead, not for mercury | primarily for lead, not for mercury | yes | no | Used ubiquitously by the holistic community, but not the best option for mercury detox. |
| ALA | use RLA | yes | yes | no | ALA passes Blood-Brain-Barrier (BBB) to detox brain |
| RLA | yes | never, makes you ill. Use ALA | yes | no | |
| Glutathione | yes, nature's way and requires liver excretion pathway support | never, too weak. More likely to move rather than cleanse mercury. | yes | no | Gets digested so it is often given intravenously (IV), delivered orally as liposomal glutathione or as precursor supplements. ACC warns against IV glutathione. |
| Glycine | ?? | yes | yes | no | Increases bile flow. |
| Milk Thistle (Sylmarin) | yes | yes | yes with meals | no | Supports liver |
| Activated Charcoal | yes. Part of the UltraBinder "catch" in the protocol. IMD thiol-functionalized silica binder preferred for mercury detox. | only during amalgam removal | yes | no | Prevent mercury re-absorption by binding to excrement |
| NBMI | no | no | no | yes | New chelator, not known by older protocols |
The ACC protocol is suspicious of weak chelators as they may bind to a mercury atom but then not maintain a hold to it in a strong enough bond to journey from the source to excretion from the body. The ACC protocol assumes that weak chelators will not maintain the bond to the mercury atom and will distribute and release mercury to other parts of the body where it may cause even more harm. It must be noted that the chelators used by the ACC protocol (DMSA, DMPS and ALA) will drop and mobilize mercury. Many of the protocols use ALA or RLA to remove mercury from the brain where mercury is causing damage and characteristics of Parkinson's disease.
There is a huge gap in the opinion about chelating with the presence of mercury amalgam fillings. ACC protocol forbids it as they believe that chelators will remove mercury from the fillings and poison your body. The ACC protocol is cautious and does not allow for the use of DMSA until amalgams are removed and forbids the use of ALA until three months after they are removed. The ACC protocol fears that since ALA can cross the "Blood-Brain-Barrier" (BBB), it might pick up mercury atoms in the body and deliver them to the brain. ACC believes in detoxifying the body first and then the brain, all after amalgams have been removed. The Dr Tom McGuire believes in the use RLA/ALA chelators before amalgam fillings are removed to lower the mercury burden as soon as possible. He does hold off on the use of DMSA until after amalgams are removed. Dr Chris Shade does not discuss the topic. Dr. Haley believes that their chelator, NBMI, does not drop mercury and thus does not have an issue with relocating mercury from amalgams.
Also see EPISODE THREE OF "THE CHELATION WARS." WHAT EXACTLY IS LIPOIC ACID?
Mixing and Combining Chelation Protocols:
The various protocols have not undergone FDA review, scientific tests or revision to optimize and improve the protocols. Each of the protocols has their pros and cons. If you are lucky enough to find a doctor knowledgeable in mercury detox you will find a full spectrum of treatment offerings from those who merely read the chelator carton directions to those who follow an existing protocol to those customize their own protocols. Dr Chris Shade can be found on video acknowledging that his "push-catch" protocol can be implemented concurrently with Dr Andy Cutler's ACC protocol. This has its merits as the "push-catch" "liver sauce" helps the liver extraction pathway and has many anti-inflammatory ingredients which will help counter the inflammation experienced from chelation agents used in the ACC protocol.
Dr. Chris Shade's YouTube video where he mentions that some practitioners overlay DMSA chelation on top of his push-catch protocol (see video 38:38 - 39:37 minutes)
Video: Dr. Christopher Shade Interview on Heavy Metal Detoxification
NBMI is a chelator for mercury without mobilization and redistribution issues, according to Dr. Boyd Halet PhD, and can be used initially to remove mercury upon which time other chelators can be employed to remove other toxic metals.
GI tract binders can be added to a protocol to help avoid reabsorption of mercury while on its way out. Binders such as QuickSilver's IMD can complement chelators such as ALA and liposomal glutathione (or its precursors NAC and glycine) which are known to excrete the body via the liver and GI tract. See below for more information on binders.
This website does not endorse chelation or any particular chelation therapy, nor with any particular experimentation of therapies.
Supplements Based Chelation:
The pharmaceutical chelators (ETDA, DMSA, DMPS, etc) are powerful heavy metal detox agents but, if misused, can have high risks of generating more damage than if never applied. Some "over the counter" supplements can act as chelators and while they have a less powerful effect, they are not immune from chelation hazards such as mercury mobilization. This is also true for high sulfur foods which are likely to stir up, mobilize and redistribute mercury to the brain.
| Supplement | Notes | |
|---|---|---|
| Garlic | study suggests that raw garlic provided protection from mercury by affecting both its assimilation and elimination.
Garlic also helps replenish sulfur body stores.
When sulfur stores are depleted, it impairs chelation effectiveness with sulfur chelators like DMPS or DMSA as they are metabolized and utilized as a source of sulfur.
Active ingredients: Allicin, Diallyl sulfide (DAS) (one sulfur atom), Diallyl disulfide (DADS) (two sulfur atoms) |

|
| Wheat Bran | study suggests that wheat bran will bind and evict mercury.
|
|
| Modified Citrus Pectin (MCP) | a gelatinous polysaccharide and soluble fruit fiber altered to be more digestible. A small study shows that it was effective in lowering blood lead levels while increasing urine lead levels.
No results shown for mercury.
Product available from EcoNugenics: PectaSol |
|
| Selenium | Found in Brazil nuts, seafood and mushrooms, selenium increases mercury excretion and decreased markers of inflammation and oxidative stress.
Selenium can also be toxic so avoid overconsumption (recommended adult allowance: 55 mcg/day, Tolerable upper limit: 400 mcg/day ref NIH)
The Physician's Desk Reference contraindicates selenium with chelators like BAL as the resulting complexes are more toxic than the metal itself, especially to the kidneys.
|

Brazil nuts are high in selenium. A serving of 3 nuts contains almost 400% daily value. Limit consumption to a serving a few times a week to avoid selenium toxicity. |
| Taurine | is a compound containing sulfur and has shown to be protective in rat studies.
|
|
| Chlorella | is an algae rich in chlorophyll, often promoted for its ability to chelate mercury and other heavy metals.
The following mouse study shows significant increases in the cumulative Hg eliminations into urine and feces.
Chlorella has been disavowed by the ACC protocol as it may cause mercury redistribution.
|
|
| Metal-X | Metal-X-Synergy is a commercial product containing a blend of many compounds (NAC, RLA, garlic, chlorella, l-glutatione, citrus pectin, etc) claimed to support the body's natural clearance of toxic metals including mercury.
Detractors of chlorella (Dr Andy Cutler) point out that it is prone to redistribute mercury and further toxify the brain and nervous system.
Note: A paper titled "Vitamin C, Glutathione, or Lipoic Acid Did Not Decrease Brain or Kidney Mercury in Rats Exposed to Mercury Vapor" (Aposhian, 2003) concluded the following:
"For the brain, NAC appeared to have increased the mercury concentration as compared to saline-treated control rats that inhaled mercury vapor" |

|
Also see:
- Medicinal plants and natural products can play a significant role in mitigation of mercury toxicity (Sanjib Bhattacharya, 2018)
Sauna Based Mercury Detoxification:
There are many sauna based heavy metal detoxification facilities and one has to be skeptical of many claims but research does support claims that sweating deserves consideration for mercury and toxic element detoxification. Sweating can also be acheived by endurance exercise which has been shown to be more effective than intensive exercise. Showering afterward can help wash-off toxins from the surface of the skin so that they are not reabsorbed. One must take care not to get dehydrated or salt, potassium and electrolyte (sodium, calcium, magnesiumm, etc) depleted as this can be hard on the kidneys. The duration must also be monitored closely and rarely exceeds 20 to 30 minutes.
Saunas take many forms, from the original Scandinavian wood lined steam room with water poured on hot rocks to far infrared (FIR), low level laser therapy (LLLT), infrared heat pads, etc. The goal is the same, sweat to excrete mercury and other toxins.
Caution: Some medications like Trihexyphenidyl (Artane) block or significantly limit the body's ability to sweat. One should avoid the use of a sauna while on this type of medication.
References:- Arsenic, Cadmium, Lead, and Mercury in Sweat: A Systematic Review (2012)
"Arsenic, cadmium, lead, and mercury may be excreted in appreciable quantities through the skin, and rates of excretion were reported to match or even exceed urinary excretion in a 24-hour period."
DOI: 10.1155/2012/184745 - Clinical response to therapeutic agents in poisoning from mercury vapor (1978)
Clinical study of a single patient cured from mercury poisoning by chelation and sauna sweat therapy.
"Analyses of the patient's sweat indicated that appreciable amounts of mercury were excreted by this route." - Could Heat Therapy Be an Effective Treatment for Alzheimer’s and Parkinson’s Diseases? A Narrative Review (2020)
DOI: 10.3389/fphys.2019.01556
- Objective Assessment of an Ionic Footbath (IonCleanse): Testing Its Ability to Remove Potentially Toxic Elements from the Body (2012)
"Contrary to claims made for the machine, there does not appear to be any specific induction of toxic element release through the feet when running the machine according to specifications."
DOI: 10.1155/2012/258968 - YouTube video: Ionic foot detox electrode scam. How it works

How the Human Body Chelates Naturally:
The human body has mechanisms to chelate mercury naturally, but it's not very effective at handling a high toxic burden. The body protects itself with the sulfur-hydrogen (SH) compounds Metallothionein and Glutathione.
Metallothionein (MT) is a cysteine-rich protein found intracellularly whuch can bind both essential and non-essential heavy metals.
Glutathione (GSH: γ-glutamyl-cysteinyl-glycine) is a natural anti-oxidant found in plants and animals. It too has a cysteine thiol group (SH) group which acts as an electron donor when binding to heavy metals (Cu, Hg, Cd, and Pb). Glutathione is poorly absorbed by the body and thus does not work well as an oral supplement. Precursors to GSH (NAC, glycine and glutamine) can be taken as supplements to suport the generation on GSH by the body. The ACC protocol recommends against the use of the GSH pathway as it mobilizes mercury and can re-absorb it in the GI tract (unless an absorbant binder is employed) while the Dr. Chris Shade protocol and some research has been in favor.
Neither are strong chelators as they form weak bonds comapared to pharmacutical chelators (eg DMSA, DMPS, ...). The half life of mercury when relying on natural, unassisted chelation by the human body, can be decades.
Note that when one is mercury toxic, that glutathione will prefer bonding with mercury, allowing a buildup of other toxic metals such as aluminum and lead. Detoxifying one's self of mercury will help in the detoxification of other toxic metals.
Also see:- Metallothionein (MT):
- Metallothionein: An overview (2007)
"MTs bind a number of trace metals including cadmium, mercury, platinum and silver, and also protect cells and tissues against heavy metal toxicity"
- Metallothionein: An overview (2007)
- Glutathione (GSH):
- Vitamin C, Glutathione, or Lipoic Acid Did Not Decrease Brain or Kidney Mercury in Rats Exposed to Mercury Vapor [cache] (Aposhian, 2003)
"The purpose of this study was to test the hypothesis that DMPS, DMSA, GSH, vitamin C, or lipoic acid, alone or in combination, would decrease brain mercury in rats exposed to elemental mercury vapor. Our results clearly indicated that none of these agents did so." - Precursors to Glutathione
- Glutathione modulation influences methyl mercury induced neurotoxicity in primary cell cultures of neurons and astrocytes (2006)
- The retention time of inorganic mercury in the brain--a systematic review of the evidence (2014)
"Estimates from modelling studies appear sensitive to model assumptions, however predications based on a long half-life (27.4 years) are consistent with autopsy findings". - Vitamin C elevates red blood cell glutathione in healthy adults (Aposhian, 2003)
"Mean red blood cell glutathione rose nearly 50% after the 500-mg vitamin C period compared with baseline"
DOI: 10.1093/ajcn/58.1.103 - Association of selenium status and blood glutathione concentrations (2011)
DOI: 10.1080/01635581.2011.535967.
- Vitamin C, Glutathione, or Lipoic Acid Did Not Decrease Brain or Kidney Mercury in Rats Exposed to Mercury Vapor [cache] (Aposhian, 2003)
Dangers of Chelation Therapy:
Essential Mineral Depletion: Chelation therapies can remove the intended "bad" heavy metals like mercury but it will also deplete the human body of essential minerals and metals. It is essential that these be reintroduced to the body as they are essential for mental health and system regulation. It may even take longer to reintroduce the required nutrients than to remove them. Thus if done improperly, chelation can result in gradual system decline and deleterious effects.
Re-distribution: The dosage and rate of intake of chelation agents is also important. Too much and the body may experience the toxic effects of mobilized mercury separated from body tissue. Dosage timing is also important as chelation agents are bonding with the mercury, but stopping at the wrong time may just dislodge the mercury without excreting it, causing the mercury to potentially relocate. One study found that chelation elevated mercury in the circulatory system which in turn elevated mercury levels in motor neuron axons across the neuro-muscular junction. Elevating the levels of mercury in motor neurons will only contribute to their decline and exacerbate Parkinson's symptoms. Mercury has a high affinity for brain and central nervous system (CNS) tissue so that mercury mobilized by chelation often finds a new home in brain and CNS tissue, harming the patient. Many popular chelation agents such as DMSA and DMPS do not cross the blood-brain-barrier (BBB) and thus can not pull mercury from the brain or central nervous system and only pull mercury from body tissue and organs. Thus mercury is pulled from body and organ tissue into the blood stream, generating a one way flow of mercury from the body to the brain and CNS.
IV and Side Effects: Intravenous (IV) chelation therapies can result in a burning sensation at the site of injection. Aggressive use of chelation agents can induce vomiting, headaches, nausea, low blood sugar symptoms and blood pressure changes.
Bad Practices: Challenge tests with a high single dosage of a chelation agent are sometimes given by clinicians to show that they can remove mercury by showing an increase of mercury in a urine test. This is often not safe as it can result in adverse effects and a spike in mercury in the system which gets relocated rather than removed.
Danger: Chelation therapy has risks including anxiety, psychosis, brain damage, liver damage, kidney damage and death. This is most often accomplished by dosages which are too high or administered over a long period of time. The chelators will mobilize mercury and will add to the mercury toxic burden in the brain. There is no way to control the random nature in which a chelator will mobilize mercury or the relocation of this mobilization. The assumption is that chelation will remove mercury over a long period of time but there is no certainty as to the benefits or damage caused during this time. Synthetic chelators are powerful drugs with the potential for serious side effects.
Saturation: Some assume that continuously chelating indefinitely, will eventually clear the body and brain of mercury. This assumption is incorrect because it relies on having the brain reach a point of saturation after which the brain will not accept any more mercury and it will eventually be cleared. It is typical that the quantity of mercury in the body is much more than that present in the brain and chelation will migrate mercury from the body to the brain. For those who are truly mercury toxic, death will be reached before the brain reaches saturation.
Kidneys: Chelation therapy using DMSA, DMPS and EDTA evict mercury out of the body via the kidneys and will relocate mercury to the kidneys potentially causing harm to the kidneys. Unlike the liver, the cells in the kidney responsible for filtering and detoxifying the blood (called nephron cells), do not regenerate. Your allocation of nephrons for your lifetime are established at birth. Chronic injury to the kidneys can lead to loss of kidney function and to end stage renal disease (ESRD). Use of chelators which excrete via the kidneys also requires that the patient increase the quantity of water consumed daily.
EDTA: It is noteworthy that EDTA comes in two forms, Na2EDTA and CaNa2EDTA. Deaths associated with chelation therapy after using Na2EDTA have been reported, related to hypocalcemia resulting in cardiac arrest. Extended use can also result in bone decalcification. The form of EDTA recommended for the chelation of toxic metals is CaNa2EDTA which does not reflect this concern.
Psychological issues: This is without a doubt the most difficult issue to navigate. Chelation can toxify the brain and nervous system with additional mercury, causing permanent harm leading to tension, anxiety, depression, the demise of one's quality of life, intolerability to their condition and suicide. Being tough and continuing to endure brain and nervous system discomfort and damage is not rewarded with a better outcome but rather a lifetime of regret. A mercury toxic brain may also fail to rationally evaluate one's condition and make poor decisions.
This website does not endorse chelation or any specific product, service, or treatment due to the extreme danger and harm that chelation can present.
Also see:- Chelation for Heavy Metals (Arsenic, Lead, and Mercury): Protective or Perilous? (2010)
DOI: 10.1038/clpt.2010.132 - Increased Inorganic Mercury in Spinal Motor Neurons Following Chelating Agents. (1996)
Study showed increased entry of mercury into motor axons across the neuro-muscular junction as a result of chelator-induced elevated circulating mercury. Chelation doubled the volume of mercury from 4% (control) to 8% of neurons.
PMID: 8856730 - Mercury binding to the chelation therapy agents DMSA and DMPS and the rational design of custom chelators for mercury
[PDF] (Graham George et al 2004)
This paper describes the faults of DMSA and DMPS and describes the bonds they make with mercury and the scientific measurements made to verify the chemistry. It also makes a case for the need for research to develop a new chelator and the scientific process which coould be employed.
"We show that neither DMSA nor DMPS forms a true chelate complex with mercuric ions and that these drugs should be considered suboptimal for their clinical task of binding mercuric ions."
"Contrary to established thinking, we show that the two functional groups of the chelator molecule cannot bind a common atom of mercury and that neither DMSA nor DMPS are well-optimized molecules for chelation therapy of mercury."
"Moreover, mercury in certain tissues (e.g., the brain) is known to be essentially impossible to remove using DMSA and DMPS ..."
"We lay out the major criteria for the rational design of metal specific or “custom chelators” and suggest directions for the development of potentially more effective drugs for mercury chelation therapy."
"We have also outlined the chemical factors that will be important in the design of a chelation therapeutic drug with specificity for mercuric ions, a custom chelator."
DOI: 10.1021/tx049904e - DMPS Backfire: The truth about DMPS - a case against "challenge tests" and DMPS
- National Capital Poison Control: Chelation: Therapy or "Therapy"?
- Mercury Poisoning - Brian Bander suicide - YouTube video
- Vitamin C, glutathione, or lipoic acid did not decrease brain or kidney mercury in rats exposed to mercury vapor (Aposhian et al, 2003)
"Specifically, we present data that GSH, vitamin C, or lipoic acid alone or in combination with DMPS or DMSA did not decrease brain mercury in rats exposed to elemental mercury." - supports the argument that chelation therapy does not work
DOI: 10.1081/clt-120022000
Binders:
Chelation therapy will grab mercury from your body, bond to it so that the resulting molecule can be evacuated by the kidneys and liver. There is the danger that when evacuated by the liver through the bile duct into the Gastrointestinal (GI) tract, that mercury can be re-absorbed in the small intestine and colon. The GI tract is about 26 feet long and gives mercury a chance to be re-absorbed into the body. This is true for any chelator which evacuates via bile and the liver, such as ALA or glutathione. To mitigate this effect, binders are often taken to absorb and attach to toxins to help the GI tract fully expel them. Binders are typically not absorbed into the bloodstream and remain in the GI tract countering the body's natural tendency to redistribute mercury. Binders can also be taken at the time of amalgam removal in case any is ingested. Note that if a binder is taken at the same time as a chelator or supplement, the binder may remove that too, thus it is common to consume a binder a half hour after a chelator. Binders should be taken on an empty stomach, between meals or in a fasted state.
Binders include activated charcoal, bentonite clay, zeolite (sodium aluminosilicate) and chitosan as well as combinations of the aforementioned substances. Activated charcoal tends to remain in the GI tract as is true of large particles of zeolite. Small "nano" particles of zeolite on the other hand will pass into the bloodstream and travel through the body and act like a chelator and will posess the same mercury redistribution pitfalls as a chelator.
Zeolite is a porous volcanic mineral with a unique and complex honeycomb crystalline structure which can trap toxins. Before using for human consumption, zeolite must be cleansed of the metals it has trapped in its cavities over the years in nature. Zeolite also uses its natural negatively charged mineral structure to draw toxins to it.
Also see:- Critical Review on Zeolite Clinoptilolite Safety and Medical Applications in vivo (2018)
- "daily intake of activated clinoptilolite suspension was effective in removal of toxic heavy metals from the body via urine"
- "great danger exists in removing the physiologically important electrolytes from the serum in a classical detoxification process, this has not been observed in clinoptilolite trials both in humans and animals..."
- "In conclusion, clinoptilolite materials tested in the scientific literature proved to be generally safe for in vivo applications even though each material seems to retain its own physical-chemical characteristics and exerts specific biological effects that cannot be readily transferable to other materials. Different particle sizes, surface areas, and cation compositions may induce different biological effects and exert different levels of effectiveness."
- Zeolite Clinoptilolite: Therapeutic Virtues of an Ancient Mineral (Mastin et al, 2019) DOI: 10.3390/molecules24081517
- Lipid Peroxidation Induced by Expandable Clay Minerals (2009)
"structural iron plays a major role in lipid peroxidation by clays", in other words, if the clay contains iron, one may experience the harmful effects of the oxidative burden caused by the iron. It's an obvious statement that clay is not bad, its what's in the clay that can be harmful.
DOI: 10.1021/es9007917
Commercial products: (not a product endorsement)
- Coseva.com - Advanced TRS: Toxin Removal System - lab-made zeolite crystal structure (not a mined mineral) for better purity with consistant particle size and pore size (0.9nm) optimized for capturing mercury.
- Waiora.com - Natural Cellular Defense (NCD) - cleansed zeolite
- Quicksilver Scientific:
- Quicksilver Scientific's IMD Binder - Intestinal Metals Detox
Contents listed as "Proprietary thiol-functionalized silica" and "Silica extract" - Quicksilver Scientific's Ultra Binder - uses activated charcoal and bentonite clay. Also includes IMD.
- Quicksilver Scientific's IMD Binder - Intestinal Metals Detox
- Results RNA ACZ Nano Zeolite - uses Clinoptilolite Zeolite as a binding agent
- ZoiGlobal: ClearDrops - Dr. Nikolas Tsirikos-Karapanos
Claims to include small particle, water soluble Zeolite so that it escapes the GI tract and passes though cellular membranes.

Quicksilver Scientific: intestinal Metals Detox (IMD) "thiol-functionalized silica" binder with an affinity for toxic heavy metals like mercury

Quicksilver Scientific: Ultra Binder for toxins (includes IMD)
Genetic Link To Deficiency To Clear Heavy Metals Like Mercury:
If one has a glutathione transferase (GST) polymorphisms, they may have a decrease in the body's ability to clear heavy metals like mercury. Patients with GST enzyme abnormalities may want to consider receiving glutathione to expedite excretion of chelated metal. Patients with the GST polymorphisms also tend to excrete mercury later in their course of treatment than other heavy metals. This can produce early false negatives for mercury, due to preferential excretion of lead and other metals. The presence of the ApoE4 mutation in the ApoE gene will also negatively affect the accumulation of mercury in one's body.
See our page on genetic links and look at SNPs rs3957357 and rs1695.
Also see:
Supplementing Your Body During Chelation:
ACC Protocol:
While DMSA and ALA may not directly remove lighter nutrient minerals, those high in mercury often experience low levels of magnesium and zinc. The ACC protocol suggests the following four "core" supplements while chelating:
| Supplement | ACC Dose | DV | Description |
|---|---|---|---|
| Vitamin C (buffered) | 1000 - 2000 mg 3X or 4X/day or 250 - 500 mg/chelator dose |
90 mg | The buffered variety is easier on the stomach and helps avoid heartburn. Dr. McGuire: 1000 mg 3X/day Warning: doses of greater than 2000 mg/day may lead to kidney stones and severe diarrhea (WebMD) |
| Vitamin E | 1,000 IU/day | 15 mg 30 IU |
Note that too much vitamin E can accumulate in body fat, cause blood thinning and bleading.
Vitamin E is an antioxidant which protexts cells from the damaging effects of free radicals.
Vitamin E refers to a group of eight compounds: alpha-tocopherol, beta-tocopherol, gamma-tocopherol, delta-tocopherol, alpha-tocotrienol, beta-tocotrienol, gamma-tocotrienol and delta-tocotrienol of which there are synthetic and natural formulations.
Gamma-tocopherol is the most prevalent form of vitamin E and is found in plant seeds, vegetable oils and nuts (pecans, almonds, sunflower seeds, walnuts).
Gamma-tocopherol has anti-inflammatory effects and is linked to a reduction in cancer and cardiovascular disease risk.
Warning: high doses can be associated with increased concentration of creatine in the urine (creatinuria) and increased risk of bleeding. |
| Magnesium-L-Thorate | 100 - 200 mg 3X or 4X/day | 420 mg | For more information see magnesium supplements
Warning: doses greater than 350 mg/day may lead to irregular heartbeat, low blood pressure, confusion, and may build up in the body causing coma and death (WebMD) |
| Zinc | 50 mg/day | 11 mg | Considered an essential mineral which can not be fabricated by the human body. Supplementation of zinc often results in copper deficiency as the two compete for absorption. Zinc supplementation may require copper supplementation (zinc/copper supplement ratio: 15/1).
Warning: high doses of 100mg/day or more for 10 or more years doubles the risk of developing prostate cancer. Doses of 10,000 mg or more can be fatal (WebMD |

The Everything Spectrum "four core" ESential 4 vitamins for chelation replenishment:
- Vitamin C: 750 mg
- Vitamin E: 268 mg
- Magnesium: 300 mg
- Zinc: 30 mg
Quicksilver Scientific (Dr. Shade):
Clear Way Cofactors is a blend of phytoextracted polyphenolic antioxidants, R-Lipoic Acid (RLA), targeted vitamins (B1, B5, B6), selenium, kelp extract and high-potency nattokinase designed to mazimize metal detoxification pathways. The botanically-sourced polyphenolics (concentrated fruit extracts and herbs: extracts of Haritaki, Pine Bark, Pomegranate, Gotu Kola, Bacopa, Dandelion Root, Bladderwrack, and Nattokinase) and RLA are included to upregulate glutathione to tackle oxidative stress, repair cellular damage and remove toxins. Clear Way Cofactors is designed to assist metal detoxification pathways and to activate the body’s antioxidant signal, called Nrf2, which regulates hundreds of genes involved in detoxification.
Clear Way Cofactors
Optional supplements:
| Supplement | ACC Dose | DV | Description and Dose |
|---|---|---|---|
| Omega 3 | 15 ml fish oil/day or 45 ml flax oil/day |
Helps with focus, concentration and mood. Dr. McGuire: 1000 mg 3X/day Warning: doses of over 3,000 mg may increase the risk of bleeding | |
| Adrenal Cortex (not Adrenal Medulla or just Cortex) | 50-250 mg 3X/day | Only needed to counter low adrenal function. See "Monitoring Your Body" in the next section.
Warning: IV injections are not considered safe. Side effects include alergic reactions, irritability and insomnia. May lower immune resistance. May have a debilitating effect on your hormonal system. | |
| Vitamin A | 25,000 IU/day | 5000 IU | Helps with immunity. Too much vitamin A can be toxic to the liver and kidneys and cause headaches and rashes.
Warning: doses greater than 10,000 (3,000 mcg) per day are generally seen as unsafe and hard on the liver. High levels of vitamin A are associated with a higher risk of osteoporosis and hip fracture (WebMD) |
| Molybdenum | 500 - 1000 mcg/day | 45 mcg | Helps prevent copper absorption. Important when taking ALA as ALA reduces copper excretion in the bile.
Warning: doses should not exceed 2,000 mcg/day. High doses may result in Gout |
| phosphatidylcholine (lecithin) | 1000 - 2000 mg/meal | Liver: Increases bile flow, help disolve cholesterol. Brain: help with poor concentration and attention deficit, depression | |
| Milk Thistle (Silymarin) | up to 750mg/day with meals | Liver: Helps with liver function by promoting glutathione production. Recommended by ACC, Dr. McGuire and Dr. Shade protocols. Dr. McGuire: 100 mg 3X/day Warning: may have estrogenic effects. May also cause an allergic reaction (anaphylaxis) (Mayo clinic) | |
| Glycine | 1000 - 3000 mg/day with meal | Liver: Helps increase bile flow. Precursor to Glutathione. Dr. McGuire: 825 mg/day | |
| taurine | 500 - 1000 mg/day with meal | Liver: Helps increase bile flow Brain: helps with anxiety. Warning: may make bipolar disorder worse (WebMD) | |
| l-tyrosine | 1000 - 3000 mg/day | Brain: help with poor concentration and attention deficit, anxiety, depression | |
| B12 | 1 - 12 mg/day | 6 mcg | Brain: help with poor concentration and attention deficit, depression.
Warning: not to be used without professional medical supervision by those who have Leber disease, megaloblastic anemia, polycythemia vera, an alergy to cobalt or cobalamin or by those who have recently received a coronary stent (WebMD) Megadosing B12 can elevate methylmercury levels!! A relationship between vitamin B12, folic acid, ascorbic acid, and mercury uptake and methylation (Zorn and Smith, 1990)
|
| Potassium | 3500 - 4700 mg | A study of the use of DMSA on children found DMSA to elevate the urinary excretion of essential minerals, especially potassium. The first round of DMSA chelation doubled the amount of potassium excreted in urine compared to the baseline. Potassium is available in bananas, fruits and vegetables. (Safety and efficacy of oral DMSA therapy for children with autism spectrum disorders: Part A - Medical results) | |
| melatonin | 3 - 50 mg | Insomnia: take one dose one hour before bed time. Available in time-release formulas. Only take if you have trouble getting to sleep.
Warning: Melatonin can make depression, diabetes, blood pressure and seizure disorders worse. May also interfere with immunosuppressive therapies (WebMD) |
Monitoring Your Body During Chelation:
It is important to monitor your body with frequent blood tests while chelating to make sure you are not damaging any organs. Dr. McGuire's guide recommends blood tests, fecal tests and a symptom evaluation every three months to monitor your detox program (chapter 8).
| Organ | Test Marker | Description | Range | Range (Quest) |
|---|---|---|---|---|
| Liver | ALT (Alanine Transaminase) | enzymes indicating damage. Heavy weight lifting can also release enzymes and elevate ALT due to muscle damage | < 41 units/L | 9 - 46 U/L |
| Liver | AST (Aspartate Aminotransferase) | enzymes indicating damage. Heavy weight lifting can also release enzymes and elevate AST due to muscle damage | < 40 IU/L | 10 - 35 U/L |
| Liver | GGT (gamma-glutamyl transferase) | liver and bile duct system. Less affected by heavy weight lifting than ALT and AST | 0 - 30 IU/L | i3 - 70 U/L |
| Liver | ALP (Alk Phos or Alkaline Phosphatase) | bile duct system | 40 - 130 U/L | 35 - 144 U/L |
| Liver | Albumin | generated by liver | 3.5 - 5.2 g/dL | 3.6 - 5.1 g/dL |
| Liver | Bilirubin | level of Bilirubin waste processed by the liver | 0.1 - 1.0 mg/dL | 0.2 - 1.2 mg/dL |
| Kidney | ACR (Albumin to Creatinine Ratio) | protein should be in blood not urine! | < 1.4 mg/dL | |
| Kidney | Creatinine | protein | 0.67 - 1.17 mg/dL | 0.70 - 1.25 mg/dL |
| Kidney | GFR (glomerular filtration rate) | calculated | 90 - 137 mL/minute | > 60mL/min/1.73m2 |
| Kidney | BUN (Blood Urea Nitrogen) | Liver releases BUN into the bloodstream, removed by the kidney and excreted in urine. If the BUN level is too high, the kidney is not functioning. A high protein diet can also elevate BUN | 8 - 20 mg/dL | |
| Thyroid | TSH (thyroid stimulating hormone) | Produced by the pituitary gland in your brain | 0.4 - 4.0 mU/L | 0.40 - 4.50 mIU/L |
| Thyroid | T3/Free T3 (triiodothyronine) | Hormone generated from T4 which regulates body temperature, heart rate and metabolism. Most of the T3 in your body is bound to protein. The T3 that isn’t bound to protein is called free T3 and circulates unbound in your blood. Total T3 is the sum of bound T3 and free T3 | Total T3: 75 - 200 ng/dL Free T3: 0.2 - 0.5 ng/dL | Free T3: 2.3 - 4.2 pg/mL |
| Thyroid | T4/Free T4 (Thyroxine) | Hormone generated by the thyroid which regulates body temperature, heart rate and metabolism. The T4 that isn’t bound to protein is called free T4 and circulates unbound in your blood. Total T4 is the sum of bound T4 and free T4 | Total T4: 4 - 11 ng/dL Free T4: 0.8-1.8 ng/dL | Free T4: 0.8 - 1.8 ng/dL |
| Adrenal gland | ACTH (Adrenocorticotropin hormone) | Generated by the pituitary gland that stimulates the adrenal glands to make cortisol. | AM: 10 - 50 pg/ml PM: 5 - 10 pg/ml | 6 - 50 pg/mL 7 - 10 am |
| Adrenal gland | DHEA-sulfate (Dehydroepiandrosterone) | Hormone which helps the body make other hormones like testosterone in men and estrogen in women and thus range is sex specific. DHEA levels drop with age and thus range is also age specific. | Male: 7 - 10 ng/mL Female: 3 - 5 ng/mL | 24 - 244 mcg/dL Age: 31 - 40 Male: 106 - 464 mcg/dL Female: 23 - 266 mcg/dL |
| Adrenal gland (cortex) | cortisol | level changes throughout the day | 0.5 - 10 ng/mL dependent on time of day | 4.0 - 22.0 mcg/dL (7 - 9 am) |
| Adrenal gland (cortex) | ALD (aldosterone) | Aldosterone balances sodium and potassium in your blood to regulate blood pressure. Sodium, potassium and chloride levels are also an indication | ng/dL | upright 8-10am: < 28 ng/dL upright 4-6pm: < 21 ng/dL supine 8-10am: 3 - 16 ng/dL |
| Blood | WBC | White blood cell count | 4.0 - 10.9 10e3 cells/mcL | 3.8 - 10.8 1000/uL |
| Blood | WBC: Neutrophil % | White blood cell differential: Neutrophil percentage Increase from baseline may indicate infection | 40 - 60 % | |
| Blood | WBC: Lymphocytes % | White blood cell differential: Lymphocytes percentage Increase from baseline may indicate infection | 20 - 40 % | |
| Blood | WBC: Monocytes % | White blood cell differential: Monocytes percentage Increase from baseline may indicate chronic inflammation | 2 - 8 % | |
| Blood | WBC: Eosinophils % | White blood cell differential: Eosinophils percentage Increase from baseline may indicate an allergic reaction | 0.2 % | |
| Blood | WBC: Basophils % | White blood cell differential: Basophils percentage | 0.5 % | |
| Blood | WBC: Neutrophil | White blood cell count: Absolute Neutrophil count (ANC) Discontinue chelating if ANC varies 1.5 from baseline: sign of neutropenia | 1.90 - 8.60 10e3 cells/mcL | 1500 - 7800 cells/uL |
| Blood | WBC: Lymphocytes | White blood cell count: Absolute Lymphocytes count | 0.53 - 4.40 10e3 cells/mcL | 850 - 3900 cells/uL |
| Blood | WBC: Monocytes | White blood cell count: Absolute Monocytes count | 0.10 - 10.0 10e3 cells/mcL | 200 - 950 cells/uL |
| Blood | WBC: Eosinophils | White blood cell count: Absolute Eosinophils count | 0.00 - 0.87 10e3 cells/mcL | 15 - 500 cells/uL |
| Blood | WBC: Basophils | White blood cell count: Absolute Basophils count | 0.00 - 0.20 10e3 cells/mcL | 0 - 200 cells/uL |
| Blood | RBC | Red blood cell count | 3.8 - 5.2 10e6 cells/mcL | 4.20 - 5.80 Million/uL |
It has been suggested in the ACC detoxification protocol that when experiencing both adrenal and thyroid problems, one should address the adrenal problems first. Adrenal support for those experiencing lethargy, can be helped by taking the supplement adrenal cortex (without adrenal medulla which contains adrenaline which can cause mood and behavior disorders).
Blood draws to test liver, kidney and thyroid function.
Monitoring Mercury Chelation Progress:
It is important to monitor how much mercury is being removed by the chelation therapy. You have reached the successful completion of treatment when the chelator is no longer removing mercury because there is no more to remove.
The ACC protocol suggests a hair test and has published a book specifically on the topic of interpreting hair analysis results. The Detoxification Manual also covers "counting rules" to help interpret the test results and to help determine if you still have toxic levels of mercury remaining. This protocol also suggests that if you are feeling the discomfort of chelation, that mercury is still being removed from your system.
Dr. Chris Shade's Quicksilver website sells a mercury "Tri-Test" which is available to doctors and tests mercury in the urine, hair and blood to determine the body's mercury burden and its ability to eliminate it. It is a unique test that measures methyl mercury and inorganic mercury separately.
The Dr. Tom McGuire protocol uses a fecal test every three months at the end of a three day chelation period. This protocol has chosen a fecal test because 90% of the eviction of mercury is through bile and the intestine. He also does not believe in employing the hair test as he feels that it measures organic methyl mercury and will not include the elemental mercury indicative of the mercury contributed by amalgam fillings. Note that it is not safe to chelate while mercury amalgam fillings are still present in one's teeth. When the mercury level tested reaches a low steady-state measurement, you have reached completion.
Neither blood, urine stool or hair tests are good at measuring the body burden of mercury lodged in mostly fatty tissue, they mearly measure the mercury circulating at any given time due to mercury ingested. The OligoScan mentioned above measures body burden directly. While the OligoScan measures mercury in one's hand, there is nothing short of an autopsy which can measure mercury in the brain and nervous system.
Also see:- The Toxicology of Mercury and its Chemical Compounds (Clarkson, Magos, Laszlo, 2006)
"... an attempt was made to calculate the lowest hair and blood levels associated with the onset of paresthesia, the first and mildest symptom of methylmercury poisoning. The investigators concluded that the onset of symptoms should occur at blood levels above 200 µg Hg/L of whole blood and above 50 µg Hg/g of hair"

Adrenal Cortex: combats chelation fatigue
Neuromelanin and Mercury:
The substance responsible for pigmentation of the skin is known to be melanin. More melanin results in darker skin. Pigmented neurons of the brain like the substantia nigra are also given their color from the presence of melanin, more specifically, neuromelanin. Studies have shown that elemental analyses of neuromelanin revealed a high sulfur content. Sulfur in turn has a high affinity for mercury. It is this sulfur affinity that chelators rely upon to bond with mercury. This affinity also draws mercury to the neurons of the substantia nigra which then interferes with the lysosomes and the process of autophagy and the reprocessing of alfa-synuclein leading to dysfunction of dopamine flow and the pathology of Parkinson's disease. Neuromelanin also has an affinity for a variety of inorganic and organic toxins.
Also see:
- Substantia nigra neuromelanin: structure, synthesis, and molecular behaviour (Zecca, Tampellini et al., 2001)
Blood Cleanse:
Cleansing or filtering mercury from blood is an option but not very effective since mercury primarily binds to body tissue and does not reside in blood. Also, mercury present as an atom and is likely too small to be trapped by a filter.
One treatment known as INUSpheresis separates blood plasma for purification and then recombines the clean plasma with with blood cells for reintroduction into the body. The treatment takes about two hours for a single cleansing session. The double filtration process removes specific antibodies, inflammatory mediators, environmental toxins, heavy metals, including mercury, and cholesterol.
This treatment is not available in the US.
Also see:
Mercury And Synergistic Toxicity:
Both mercury and aluminum are accepted to be neurotoxins but when the two act together they are much more neurotooxic than if they acted alone.
Also see:
- Synergism in aluminum and mercury neurotoxicity (Alexandrov et al, 2018)
"aluminum sulfate and mercury sulfate when added together exhibit a remarkable and significant synergism in their activation of the NF-kB (p65) complex to manifold than that which each metal sulfate could induce by itself"
A Mercury Amalgam Fillings Data Point:
This is a screen capture image from the Michael J. Fox 1994 movie "Greedy" which shows that he has a mouthfull of Silver-Mercury dental amalgam fillings. This is taken at 28 minutes and 12 seconds into the movie. Coincidence? We don't think so.
Also see:
- IMDB: Greedy (1994)
info and trailer. - YouTube movie clip showing silver-mercry amalgam fillings



Chelation Pros:
- The extraction of mercury from the human body will help reduce its neurotoxic effects. Unfortunately there is no effective chelation therapy nor is there anyone working on the problem.
- The source of mercury exposure can be avoided. Mercury dental amalgams can be removed safely and are often the primary source of intoxication. Avoiding dietary sources of mercury, such as fish, will help reduce future bioaccumulation.
Chelation Cons:
- While some chelation agents are determined to be safe by the FDA, the chelation agents can invoke an allergic reaction, nausea, dizziness, headache and discomfort in some patients. Chelation agents bind to mercury and mobilize it so that the kidneys and liver can excrete it. The kidneys and liver can also be damaged during the process. There is always the danger that some chelation agents (EDTA, DMSA, DMPS, ALA) will "drop" the mercury in an undetermined and unexpected part of the body including motor neurons where it may cause new damage. Mercury has an affinity for brain and central nervous system (CNS) tissue and chelation may mobilize mercury from low risk areas of the body to high risk and high misery inducing areas such as the brain and CNS. High levels of chelation can disrupt and disperse enough mercury to be considered toxic. There is also very little research on the successful evacuation of mercury from the brain and CNS by chelators. Much more research is needed.
- Chelators which can not pass through the blood-brain-barrier (DMSA, DMPS, etc) will not be able to remove mercury from the brain and CNS. These chelation agents tend to pull mercury from body tissue and organs, release it into the circulatory system where mercury can navigate to the brain and CNS, causing serious damage.
- There is no good way to measure one's mercury burden. Blood, urine, stool and hair tests are useless when attempting to measure long term, chronic exposure. The light spectroscopy based Oligoscan can only measure mercury in hand tissue as a proxy and it gets no respect from the medical community.
Also see the our PDCure.org project to develope a new mercury chelator.